Acquires intellectual property to support pursuit of regulatory approval for innovative treatment
TORONTO, May 26, 2022 /PRNewswire/ – Minimally invasive image-guided therapy start-up, SpinaFX Medical Inc. (SpinaFX), today announced significant milestones hit since closing its $6.88M CDN Series A round in November 2021. SpinaFX has leveraged investment proceeds for patent estate expansion, product enhancements, quality management system implementation, regulatory approval pursuits and commercialization of Triojection®.1
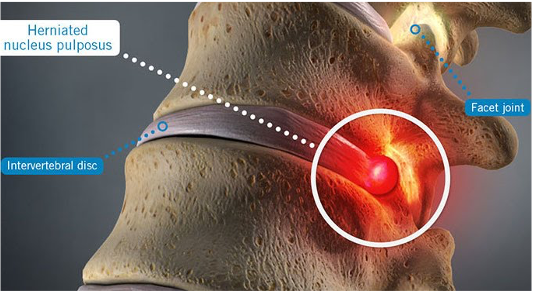
SpinaFX acquired the world’s largest collection of patents, trademarks, pre-clinical and clinical evidence, and regulatory and commercial documentation surrounding intradiscal oxygen/ozone therapy for treating lumbar disc herniations. Intradiscal oxygen/ozone therapy involves the injection of a mixture of oxygen and ozone gas into an intravertebral contained herniated disc for relief of leg and back pain.
The acquisition of existing IP yielded a powerful repository of evidence for intradiscal ozone injection involving bench, animal and randomized clinical trial data, as well as 65 patents covering the generation, measurement, delivery, and clinical use of technology for intradiscal ozone injection. SpinaFX is now well positioned to advance its products and accelerate regulatory approval in Canada, the EU, the U.S. and beyond.
“There is an indisputable gap within the continuum of care for contained herniated lumbar discs,” said Jeff Cambra, SpinaFX CEO. “I am extremely pleased with the confidence placed in the SpinaFX team by investors and to have acquired the collective knowledge of the most impressive companies behind intradiscal ozone technology under the umbrella of SpinaFX. We are moving decisively to bring this technology to market to address the unmet needs of patients and providers.”
Since closing the funding round in November 2021, SpinaFX has achieved several additional key milestones, further positioning the Company to achieve its goal of bringing innovative, minimally invasive image-guided treatments to the global market:
- Built a world-class leadership team, including leading interventional neuroradiologist Dr. Kieran Murphy as Chief Medical Officer
- Acquired existing device finished goods and manufacturing inventory
- Partnered with StarFish Medical for commercialization contract manufacturing support
“With these investments and acquisitions, SpinaFX now has the initial capital needed to commercialize Triojection® and the IP base to protect our competitive advantage for future expansion,” said Igor Keselman, SpinaFX Executive Chair.
The Company looks forward discussing these accomplishments with leading interventional radiologists as an exhibitor at the Society of Interventional Radiology 2022 Annual Meeting in June. To learn more about SpinaFX, visit SpinaFX.com.
About SpinaFX
Headquartered in Ontario, Canada, SpinaFX is a medical device company bringing innovative, minimally invasive image-guided treatments to patients with back pain due to herniated discs.
Forward-Looking Statement
This news release contains predictions, expectations, beliefs, plans, projections, objectives, assumptions or future events or performance, referred to herein as “forward-looking statements”, which are made as of the date of this news release or as of the effective date of information described in this release, as applicable. The forward-looking statements address such anticipated events or occurrences which may include economic factors, industry trends, market demand, and corporate performance and profitability. Forward-looking statements are often identified through words or expressions including “expects”, “anticipates”, “plans”, “projects”, “estimates”, “envisages”, “assumes”, “intends”, “strategy”, “goals”, “objectives” or variations thereof or stating that certain actions, events or results “may”, “can”, “could”, “would”, “might” or “will” be taken, occur or be achieved, or the negative of any of these terms and similar expressions.
All forward-looking statements are based on current beliefs as well as various assumptions made by, and information currently available to SpinaFX’s management team. By their very nature, forward-looking statements involve inherent risks and uncertainties, both general and specific, and risks exist that estimates, forecasts, projections, and other forward-looking statements will not be achieved or that assumptions do not reflect future experience. We caution any person reviewing this news release not to place undue reliance on these forward-looking statements as a number of important factors could cause the actual outcomes to differ materially from the beliefs, plans, objectives, expectations, anticipations, estimates assumptions and intentions expressed in such forward-looking statements.
Neither SpinaFX nor its representatives undertake to update any forward-looking statement, whether written or oral, that may be made from time to time by SpinaFX or its representatives or on behalf of either of them, except as may be required by law.
| 1 | Triojection® Intradiscal Ozone injection therapy from SpinaFX is not currently approved for sale by regulatory agencies in the U.S., Canada, EU or elsewhere. |
SOURCE Spinafx medical inc