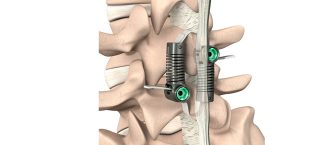
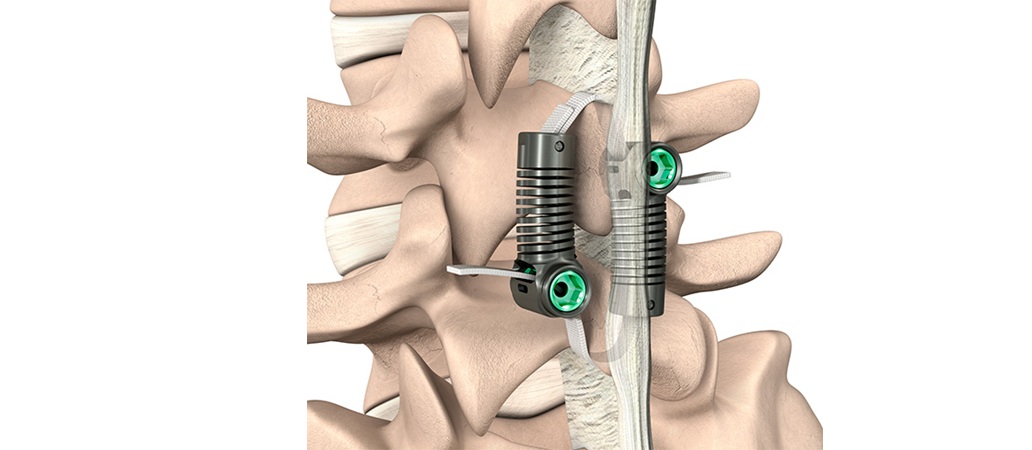
Smith+Nephew Announces First Clinical Cases with the Next-Generation CORI◊ XT Handheld Robotics Platform Across Knee and Shoulder Arthroplasty
Smith+Nephew (LSE: SN, NYSE: SNN), a global medical technology company, announces the completion of the first clinical cases performed using the next-generation CORI◊ XT Handheld Robotics…