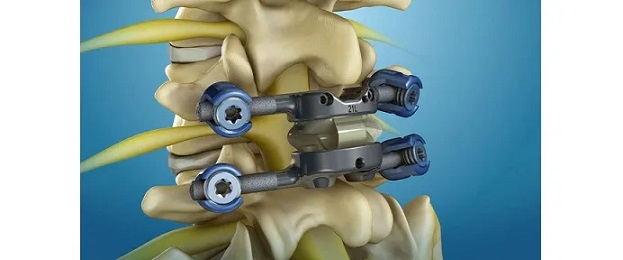
SALT LAKE CITY, Oct. 11, 2023 (GLOBE NEWSWIRE) — SINTX Technologies, Inc. (www.sintx.com) (NASDAQ: SINT) (“SINTX” or the “Company”), an original equipment manufacturer of advanced ceramics, announced today it has been awarded a Phase II grant of $1,972,826 by the National Institutes of Health (NIH) to develop a 3D printed (3DP) composite silicon nitride – polyetheretherketone (SN-PEEK) spinal implant. This represents the fourth NIH grant awarded to SINTX since the Phase I award for this project was received in September of 2021.
Representing approximately 40% of spine implants within the $10 billion global spine market, PEEK-based devices are favored because of their proven clinical efficacy. Through the development of 3DP SN-PEEK spinal implants, SINTX aims to combine the familiarity and benefits of PEEK with the antibacterial and osteoconductive characteristics of silicon nitride. These composite implants will be manufactured with modern, cost-effective 3D printing technologies.
“Our successful completion of Phase I aims has shown the feasibility and reliability of 3DP SN-PEEK spinal implants. These 3DP implants met minimum load thresholds in accordance with established mechanical testing standards, demonstrated basic biocompatibility, and resisted bacterial adhesion. These findings established the foundation for this Phase II Small Business Innovation Research (SBIR) award,” said Dr. Ryan Bock, SINTX Vice President of Research and Development and Principal Investigator for the project. “In Phase II, SINTX and external collaborators will conduct formal design validation activities; test static and dynamic mechanical properties of implants 3DP according to the revised design; carry out formal biocompatibility assessments; and assess in vivo antimicrobial activity, biocompatibility, and osseointegration for a 3DP SN-PEEK cervical implant against appropriate controls.”
SINTX will collaborate with the University of Pennsylvania School of Veterinary Medicine (Penn Vet) over the next 24 months to execute this project. The NIH grant will support resourcing and acquisition of materials as well as testing and trials at SINTX and Penn Vet. Upon the successful execution of the Phase II grant application goals, SINTX will have the majority of the benchtop and large animal in vivo data needed to seek FDA regulatory clearance using the 510(k) pathway. Additionally, successful demonstration of infection prevention in vivo will be a critical step towards enabling an antimicrobial device claim. These preliminary animal data will be used to seek future funding from NIH or other Federal agencies to further validate antimicrobial properties of devices made from SN-PEEK.
“In the Phase I effort, Dr. Steven Kurtz of Drexel University and Dr. Noreen Hickok of Thomas Jefferson University were the lead university investigators while Dr. Thomas Schaer of Penn Vet provided valued consultative support for study and implant design. These scientists provided significant contributions to the proposal, and we are grateful for their continued support,” said Dr. Sonny Bal, President, and CEO. “Dr. Schaer will be the lead investigator at Penn Vet while Drs. Kurtz and Hickok will assume advisory roles on the project during execution of Phase II activities. Since the NIH grant will fund the acquisition of needed benchtop data and the first ever in vivo performance data for SN-PEEK devices, the award represents a critical milestone in expanding the antibacterial and osseointegrative properties of silicon nitride into composite structures. From a commercial standpoint, this NIH-funded project is an important step in our strategy to introduce a new generation of highly differentiated implants into the spine market.”
Research reported in the “3D Printed Silicon Nitride Porous PEEK Composite Spinal Cages for Anti-Infection” proposal was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health under award number 2R44AR083836-02.
The content included in this release is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
About SINTX Technologies, Inc.
SINTX Technologies is an advanced ceramics company that develops and commercializes materials, components, and technologies for medical and technical applications. SINTX is a global leader in the research, development, and manufacturing of silicon nitride, and its products have been implanted in humans since 2008. Over the past two years, SINTX has utilized strategic acquisitions and alliances to enter into new markets. The Company has manufacturing facilities in Utah and Maryland.
For more information on SINTX Technologies or its materials platform, please visit www.sintx.com.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 (PSLRA) that are subject to a number of risks and uncertainties. Risks and uncertainties that may cause such differences include, among other things: development of SN-PEEK spinal implant products may take longer than expected and such products may not prove to be as effective as other products currently being commercialized or to be commercialized in the future by competitors; risks inherent in manufacturing and scaling up to commercial quantities while maintaining quality controls; volatility in the price of SINTX’s common stock; the uncertainties inherent in new product development, including the cost and time required to commercialize such product(s); market acceptance of our products once commercialized; SINTX’s ability to raise funding and other competitive developments. Readers are cautioned not to place undue reliance on the forward-looking statements, which speak only as of the date on which they are made and reflect management’s current estimates, projections, expectations and beliefs. There can be no assurance that any of the anticipated results will occur on a timely basis or at all due to certain risks and uncertainties, a discussion of which can be found in SINTX’s Risk Factors disclosure in its Annual Report on Form 10-K, filed with the Securities and Exchange Commission (SEC) on March 29, 2023, and in SINTX’s other filings with the SEC. SINTX disclaims any obligation to update any forward-looking statements. SINTX undertakes no obligation to publicly revise or update the forward-looking statements to reflect events or circumstances that arise after the date of this report.
Business and Media Inquiries for SINTX:
SINTX Technologies 801.839.3502
IR@sintx.com
Source/Image: SINTX Technologies, Inc.