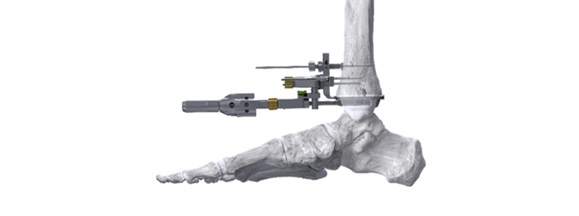
ENGLEWOOD, Colo., September 10, 2024–(BUSINESS WIRE)–Paragon 28, Inc. (NYSE: FNA) is pleased to announce the addition of a novel Right-Angle Drill to the APEX 3D™ Total Ankle Replacement System designed to improve tibia preparation prior to the implantation of the APEX 3D™ tibia implant. The Right-Angle Drill utilizes a linear guide to precisely drill vertical holes into the tibia for ideal peg placement and a highly stable tibia implant interface.
The release of the Right-Angle Drill continues a series of instrumentation enhancements to the APEX 3D™ Total Ankle Replacement System, which includes the recently released Polishing Blocks, Square Tip Drill, and next-generation Off-Set Impaction Handle. These recent enhancements were primarily designed to improve tibia preparation, thereby contributing to the long-term success of ankle replacements and ultimately improving surgeon experience and patient outcomes.
“The preparation of the tibia prior to placing the implant is paramount for long-term success. Successful total ankle surgery is characterized by an implant bone interface, prepared tibia bone and the tibia implant, which combine into one structure that works together,” says Dr. Mark Myerson, a design surgeon of the APEX 3D™ system. “The Right-Angle Drill precisely prepares the tibia to create the appropriate interface for the APEX 3D™ implant to allow for long-term survival.”
“We are thrilled to continue advancing our APEX 3D™ Total Ankle Replacement System with the launch of our novel Right-Angle Drill,” says Albert DaCosta, CEO and Chairman of Paragon 28.“The Right-Angle Drill is a critical step towards addressing tibia implant loosening, the primary cause of total ankle replacement failure. This drill enables surgeons with a more precise preparation and promotes better implant integration, which we believe will result in improved long-term outcomes for foot and ankle patients.”
Paragon 28 will be showcasing the APEX Right-Angle Drill at the American Orthopaedic Foot & Ankle Society (AOFAS) Annual Meeting this week in Vancouver, British Columbia. Additionally, Paragon 28 will be showcasing the recently launched R3FLEX™ Stabilization System, SMART28℠ Case Management Portal, SMART Bun-Yo-Matic℠, FJ2000™ Power Console and Burr System, and PRECISION® MIS Bunion System throughout the meeting. Surgeons may visit booth 625 or schedule a hands-on introduction to these technologies with P28 product experts who will be on-site throughout the meeting.
About Paragon 28, Inc.
Based in Englewood, CO., Paragon 28, is a leading medical device company exclusively focused on the foot and ankle orthopedic market and is dedicated to improving patient lives. From the onset, Paragon 28® has provided innovative orthopedic solutions, procedural approaches and instrumentation that cover a wide range of foot and ankle ailments including fracture fixation, forefoot, ankle, progressive collapsing foot deformity (PCFD) or flatfoot, Charcot foot and orthobiologics. The company designs products with both the patient and surgeon in mind, with the goal of improving outcomes, reducing ailment recurrence and complication rates, and making the procedures simpler, consistent, and reproducible.
Forward Looking Statements
Except for the historical information contained herein, the matters set forth in this press release are forward-looking statements within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995, including, but not limited to: Paragon 28’s potential to shape a better future for foot and ankle patients. You are cautioned not to place undue reliance on these forward-looking statements. Forward-looking statements are only predictions based on our current expectations, estimates, and assumptions, valid only as of the date they are made, and subject to risks and uncertainties, some of which we are not currently aware. Forward-looking statements should not be read as a guarantee of future performance or results and may not necessarily be accurate indications of the times at, or by, which such performance or results will be achieved. These forward-looking statements are based on Paragon 28’s current expectations and inherently involve significant risks and uncertainties. Actual results and the timing of events could differ materially from those anticipated in such forward-looking statements as a result of these risks and uncertainties. For a further description of the risks and uncertainties that could cause actual results to differ from those expressed in these forward-looking statements, as well as risks relating to Paragon 28’s business in general, see Paragon 28’s current and future reports filed with the Securities and Exchange Commission, including its Annual Report on Form 10-K/A for the fiscal year ended December 31, 2023 and its Quarterly Reports on Form 10-Q, as updated periodically with its other filings with the SEC. These forward-looking statements are made as of the date of this press release, and Paragon 28 assumes no obligation to update the forward-looking statements, or to update the reasons why actual results could differ from those projected in the forward-looking statements, except as required by law.
Disclaimer
Dr. Myerson may report consulting and royalty fees from Paragon 28 in connection with the provision of product development services to Paragon 28.
Nothing in this material is intended to provide specific medical advice or to take the place of written law or regulations.
Contacts
Investor Contact
Matthew Brinckman
Senior Vice President, Strategy and Investor Relations
Phone: (720) 912-1332