FREMONT, Calif., Nov. 19, 2024 /PRNewswire/ — THINK Surgical, Inc., an innovator in the field of orthopedic surgical robots, today announced that its TMINI® Miniature Robotic System has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for use with the Freedom Total Knee, Freedom Titan Knee, and Freedom Primary PCK Systems from Maxx Orthopedics, Inc. (Norristown, PA) under a Collaboration Agreement between the two companies.
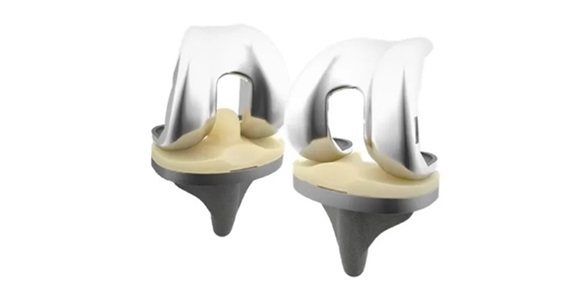
The Freedom Total Knee, Freedom Titan Knee, and Freedom Primary PCK Systems have been added to THINK Surgical’s ID-HUB™, a proprietary data bank of implant modules for use with the open platform version of its TMINI System in the United States.
The Freedom Total Knee and Freedom Titan Knee Systems feature a multi-radius design that accommodates individual patient anatomy, potentially leading to improved alignment and positioning for long-term success. It offers an enhanced range of motion that can achieve flexion up to 155 degrees to help improve functional outcomes for active lifestyles after surgery. Clinically, it has demonstrated strong performance and durability with a 10-year survivorship of 98.3%1.
“Maxx Orthopedics believes in great partnerships and is excited surgeons will have the ability to perform robotic-assisted TKA procedures with our Freedom Total Knee System. Combining our knee solutions with the TMINI System, surgeons can choose from multiple implant options- standard materials to metal sensitivity solutions- and seamlessly perform conventional or robotic-assisted procedures,” said Ashesh Shah, chief executive officer of Maxx Orthopedics.
“THINK Surgical is excited to offer the TMINI System for use with the Freedom Knee System. The open platform version of TMINI is now available with eight different knee designs from eight separate orthopedic companies offering surgeons the choice of single-radius, anatomic (multi-radius), and guided motion knees on a single robotic platform” said Stuart Simpson, president, and chief executive officer of THINK Surgical.
References
1. Durbhakula S, Rego L, Eberle R: Restoration of Femoral Condylar Anatomy for Achieving Optimum Functional Expectations: Continuation of Earlier Studies at 10-Years follow-up. J Orthop Exp Innovation, [Accepted for Publication], 2024.
About THINK Surgical, Inc.
THINK Surgical, Inc., is a privately held U.S.-based technology innovator that develops and markets orthopedic robots.
THINK Surgical actively collaborates with healthcare professionals to refine our orthopedic products, improving the lives of those suffering from advanced joint disease with precise, accurate, and intelligent technology. Please refer to the instructions for use for the TMINI Miniature Robotic System for a complete list of indications, contraindications, warnings, and precautions. For additional product information, please visit www.thinksurgical.com.
THINK Surgical and TMINI are trademarks of THINK Surgical, Inc.
About Maxx Orthopedics
Maxx Orthopedics is the manufacturer of the Freedom Total Knee System, Freedom Titan Knee System, Freedom PCK System, Libertas® Total Hip System, and Quick Recovery Solutions (QRS®). The company develops and markets innovative orthopedic medical devices worldwide with a focus on providing state-of-the-art implants and related solutions that best restore patient mobility while accommodating lifestyle, anatomical and economic needs. Discover more at www.maxxortho.com/ and follow us on LinkedIn, X, Instagram, and Facebook.
Media Contacts:
THINK Surgical Maxx Orthopedics, Inc.
Nick Margree Donald Montgomery
nmargree@thinksurgical.com info@maxxortho.com
SOURCE THINK Surgical, Inc.