Facilitates Least Invasive All-Inside, No Metal ACL Reconstruction
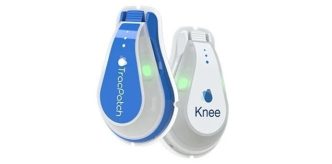
NAPLES, Fla., Feb. 26, 2026 /PRNewswire/ — Arthrex, a global leader in minimally invasive surgical technology, announced that it has released the TightRope® SB implant, the first device on the market for anterior cruciate ligament (ACL) reconstruction using all-suture, soft-button technology. To date, nearly two thousand implants have been used in procedures across the U.S.
ACL reconstruction is one of the most performed orthopedic procedures worldwide, helping patients across all activity levels—from elite athletes to active adults—return to stability, confidence and movement. The TightRope® SB implant represents the evolution of Arthrex ACL technology, offering a fully all‑suture solution while maintaining the procedural flexibility and reproducibility surgeons expect.
By eliminating rigid materials and preserving strong, adjustable fixation, the device expands graft-fixation options for both ACL reconstruction and repair. This less invasive approach supports improved knee stability and range of motion,1,2 while eliminating the presence of metal implants.
“We look forward to seeing how patients benefit from this revolutionary solution for ACL reconstruction,” said Arthrex Knee Ligament Senior Group Product Manager Justin Boyle. “The TightRope SB builds on Arthrex’s foundation of knee ligament innovation and reflects how ACL surgery continues to evolve.”
The all-suture, soft-button technology used in the TightRope SB implant offers high-strength fixation and features a flat-tape loop that holds the new ligament in place.2 The implant is also radiopaque, which helps surgeons confirm its position during and after surgery. The implant is available in a variety of configurations to support ACL reconstruction using all graft types as well as ACL primary repair procedures.
CU Anschutz Orthopedics and UCHealth surgeon Rachel M. Frank, MD (Aurora, Colorado), completed the first cases with the TightRope SB implant in June 2025. Both cases were QuadLink™ all-inside ACL reconstructions with the InternalBrace™ technique.
“The TightRope SB implant introduces a metal-free, screw-free option for ACL reconstruction fixation,” said Dr. Frank. “The adjustable, all-suture construct provides intraoperative flexibility in graft tensioning and a low-profile footprint. Together, these design features reflect an important step forward in ACL fixation, with continued evaluation underway to assess clinical outcomes.”
In April 2025, the TightRope SB implant received gold in the Healthcare Environment and Patient Care Solutions category at the 38th annual Edison Awards.
“Winning gold at the Edison Awards is a powerful validation of our shared commitment to innovation and our mission of Helping Surgeons Treat Their Patients Better®,” said Boyle. “This recognition reflects the purpose‑driven design behind TightRope SB and its role in advancing ACL reconstruction.”
For more information, downloadable multimedia assets and interview requests for subject matter experts, contact Arthrex Media Relations.
About Arthrex
Arthrex, headquartered in Naples, Florida, is a global medical device company and leader in multispecialty minimally invasive surgical technology innovation, scientific research, manufacturing and medical education. The company has pioneered the field of arthroscopy and sports medicine and develops more than 1,000 new products and related procedures annually to advance minimally invasive surgery for orthopedics, trauma, and spine, in addition to arthroplasty, cardiothoracic, and biologic innovations. Arthrex also specializes in the latest 4K multispecialty surgical visualization and OR integration technology solutions. For more information, visit Arthrex.com.
References
- van der List JP, DiFelice GS. Primary repair of the anterior cruciate ligament: A paradigm shift. Surgeon. 2017;15(3):161-168. doi:10.1016/j.surge.2016.09.006
- Arthrex, Inc. Data on file (APT-07399). Naples, FL; 2025.
The InternalBrace surgical technique is intended only to augment the primary repair/reconstruction by expanding the area of tissue approximation during the healing period and is not intended as a replacement for the native ligament. The InternalBrace technique is for use during soft tissue-to-bone fixation procedures and is not cleared for bone-to-bone fixation.
SOURCE Arthrex, Inc.