Proven Solution, Greater Heights – Now with Trochanteric Entry
Orthofix – March 4, 2026 – We are pleased to announce the commercial launch of the Fitbone™ Trochanteric Lengthening System in the United States, marking a significant expansion of the Fitbone internal limb lengthening portfolio. This latest addition introduces a trochanteric entry option designed to support both adult and pediatric patients and broaden the clinical possibilities in modern limb reconstruction.
Advancing Internal Limb Lengthening Technology
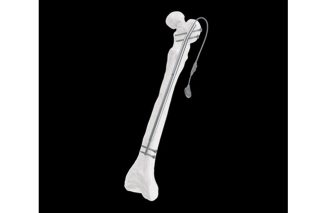
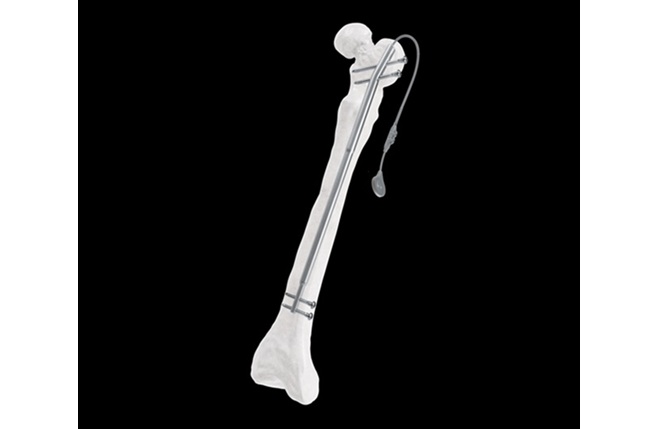
The Fitbone™ Trochanteric Lengthening System brings forward a fully implantable solution for femoral lengthening that supports a bone‑conserving, minimally invasive approach. Featuring the smallest proximal diameter currently available, it offers an important advancement for treating pediatric and small‑stature patients (ages 12–21) who require a safer entry point through the greater trochanter.
Grounded in more than 20 years of surgical experience, Fitbone continues to pair regenerative principles with implantable technology that respects alignment, anatomy, and soft tissues throughout the procedure.
What’s New in the Expanded Fitbone™ System
The trochanteric entry configuration adds meaningful versatility to the Fitbone portfolio:
- Smaller diameter options to support pediatric and small stature patients
- Extended lengthening ranges that broaden treatment possibilities
- Purpose‑designed instrumentation for limb alignment and soft tissue protection
- Trial nails that assist with safe trajectory assessment and blocking screw placement
This expansion enables surgeons to address a wider spectrum of femoral lengthening needs while maintaining the controlled, predictable distraction mechanics that have long defined the Fitbone platform.
Clinical Applications in Limb Reconstruction Surgery
The system is intended for the correction of limb length discrepancies and supports a variety of reconstruction needs across pediatric and adult patients. With an antegrade approach through the greater trochanter, the new design provides an entry point that aligns well with the anatomical requirements of smaller femoral canals—an essential consideration for younger or small stature individuals.
The trochanteric option complements existing Fitbone solutions, including:
- Fitbone™ TAA Femoral and Tibial Nails
- Fitbone™ Transport and Lengthening System, used for treating bone defects arising from trauma, infection, or tumor‑related conditions
Together, these systems form an intramedullary portfolio tailored to treat limb length discrepancies and complex bone defects.
Benefits of Internal Limb Lengthening with Fitbone™
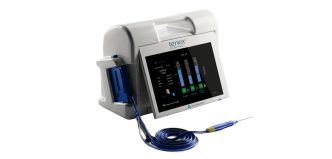
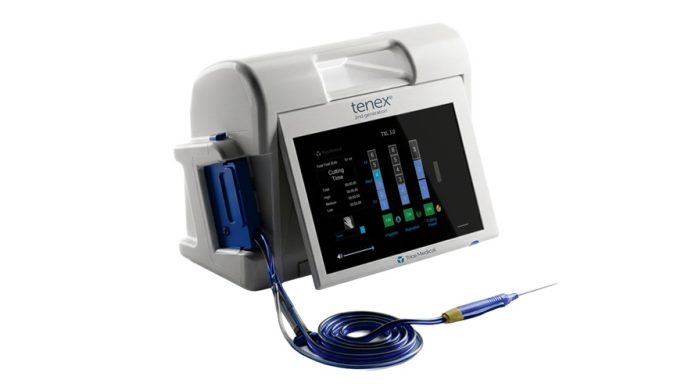
The Fitbone™ Trochanteric Lengthening System retains the core electromagnetic technology that has delivered consistent clinical reliability for more than 25 years. Key advantages include:
- Direct power delivery to the nail, ensuring dependable energy transmission
- Consistent distraction force
- Energy transmission unaffected by patient size or soft‑tissue thickness
- Lengthening sessions completed in approximately 90 seconds
- Compact, easy‑to‑use Control Set with no magnets and no special alignment requirements
These performance characteristics help create a predictable, controlled lengthening process—an essential component of successful distraction osteogenesis.
FAQs on Limb Lengthening Systems
Who is the Fitbone™ Trochanteric Lengthening System designed for?
It supports both adult patients and pediatric patients ages 12–21, especially those requiring a smaller proximal diameter or a trochanteric entry approach.
Does the system replace existing Fitbone technologies?
No. It complements the Fitbone TAA and Fitbone Transport and Lengthening systems, expanding treatment options within the portfolio.
What makes the technology reliable?
The Fitbone platform is built on more than two decades of clinical use and features a direct electromagnetic drive mechanism that maintains consistent distraction force, independent of nail type or patient size.
Learn More About Fitbone Trochanteric Lengthening System
Fitbone™ is a trademark or registered trademark of Orthofix Medical Inc., or its affiliated companies.