Label expansion positions SINEFIX for wider adoption in rotator cuff repair.
Broader indications reinforce SINEFIX as a versatile alternative to traditional suture anchors for soft tissue-to-bone fixation.
MINNEAPOLIS, April 21, 2026 /PRNewswire/ — Inovedis Inc., a cutting-edge orthopedic company, announced today that the U.S. Food and Drug Administration (FDA) has cleared expanded clinical applications for the SINEFIX Implant. Designed to replace traditional suture anchors for soft tissue-to-bone fixation, SINEFIX can now be used across a wider range of procedures, including:
- Shoulder: Rotator Cuff Repairs, Deltoid Repair.
- Foot/Ankle: Achilles Tendon Repair.
- Knee: Medial Collateral Ligament Repair, Lateral Collateral Ligament Repair, Patellar Tendon Repair, Quadriceps Tendon Rupture Repair.
- Elbow: Biceps Tendon Reattachment, Tennis Elbow Repair, Ulnar or Radial Collateral Ligament Reconstruction, Lateral Epicondylitis Repair.
- Hip: Gluteus Medius Repair, Gluteus Minimus Repair.
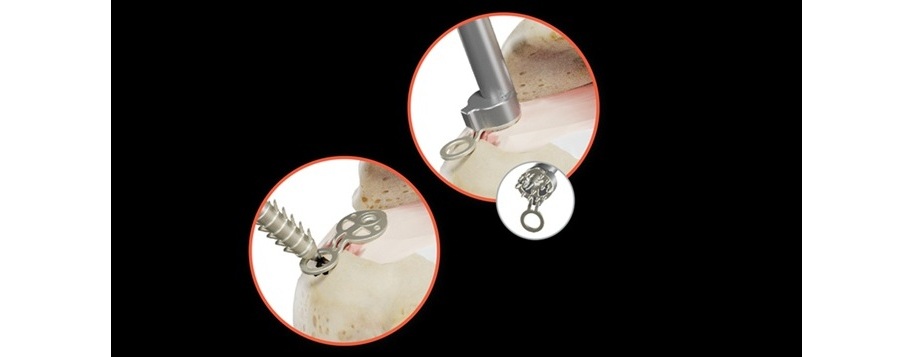
SINEFIX is composed of a polyether ether ketone (PEEK) and includes one baseplate and two PEEK anchors, equipped with additional teeth to prevent tendon slippage. The innovative design addresses key limitations of traditional fixation methods.
Rather than relying on sutures that can concentrate force and cut through tendon, SINEFIX secures tissue by distributing compression across a broad surface area, effectively “stapling” the tendon to bone. This approach eliminates point-loading of sutures and distributes the pressure evenly across the repair footprint while also maintaining blood circulation at the repair site, which is critical to biological healing.
“Using SINEFIX for gluteal repairs cuts down the procedure time by one-third compared to a standard repair since there is no suture sorting. It is far faster and better than the standard technique,” stated Christopher Dougherty, DO. “Plus, the biggest advantage is that you don’t have to disrupt the tendon to fix it unlike other repair methods. Ideally, the goal is to preserve the tendon.”
According to Michael R. Redler, MD, “SINEFIX is a really exciting new technology that addresses the many challenges with rotator cuff repair. Its use with TendoGrip allows for controlled tensioning and improved footprint coverage in a streamlined approach, without the complexity of suture management. No other system lets you tension and insert in one step. Expanding its indication across all tear sizes further enhances its utility in soft tissue repair.”
“We are extremely happy to be able to meet the demand from surgeons to utilize SINEFIX in these extended indications,” said Lukas Floess, CEO and Co-founder. “Surgeons are no longer limited to using SINEFIX for small rotator cuff tears, and the prospects for its potential adoption in other areas of the body are considerable. This is a significant achievement for the company, and we look forward to advancing this technology in other areas of sports medicine and orthopedics.”
ABOUT INOVEDIS
Inovedis is a cutting-edge medical technology company dedicated to offering innovative solutions that improve patient outcomes while minimizing the complexity of surgical interventions. SINEFIX™, their flagship product, was designed to transform rotator cuff repair by addressing both biomechanical fixation and the critical role of blood flow in tendon-to-bone healing.
With newly cleared clinical indications spanning the shoulder, hip, knee, elbow, and foot & ankle, SINEFIX expands its reach across a wide range of orthopedic and sports medicine applications.
SINEFIX is approved for sale in the U.S.
To learn more, visit our website and follow us on LinkedIn.
MEDIA CONTACT:
Ronda Taylor
info@inovedis.com
SOURCE Inovedis