Next-generation platform now offers deeper tissue access and enhanced procedural efficiency for physicians
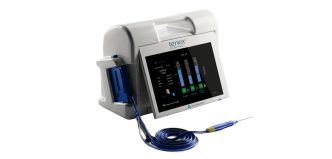
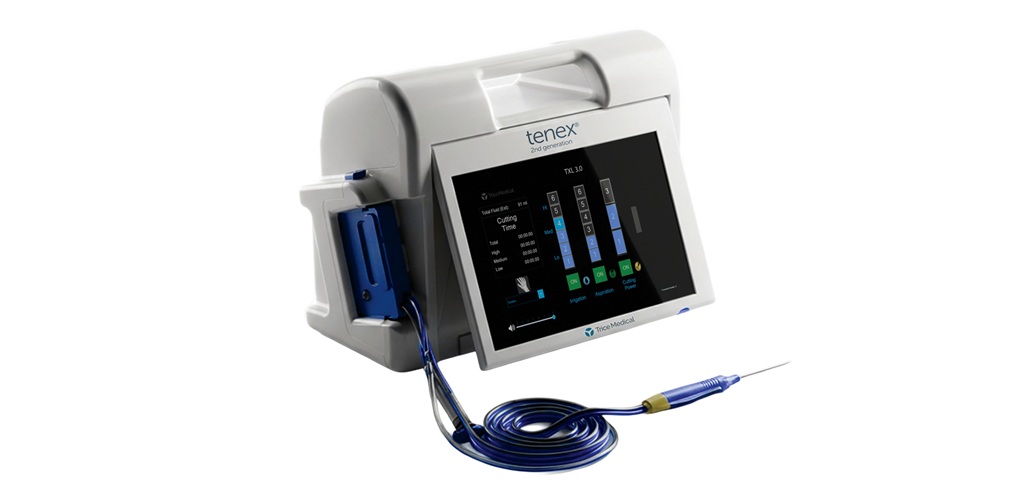
MALVERN, Pa., March 17, 2026 -(BUSINESS WIRE)- Trice Medical today announced nationwide commercial availability of the FDA-cleared Tenex® 2nd Generation System, building on the proven Tenex Health TX® platform that has been used in more than 240,000 procedures.
The Tenex 2nd Generation System is an advanced platform designed to expand minimally invasive ultrasonic procedures for the treatment of chronic tendon disease and other musculoskeletal conditions, including calcific deposits, bone spurs, and diabetic foot ulcers.
Tenex 2nd Generation introduces two longer versions of the ultrasonic handpiece (MicroTips), enabling physicians to reach deeper anatomical structures and improve access to areas such as the shoulder and hip. The system also provides advanced power settings and expanded aspiration and irrigation controls through a redesigned console.
“We are proud to announce the successful completion of the limited-use release of our Tenex 2nd Generation System and are excited to make this enhanced platform available to a broader market,” said Mike Steen, CEO of Trice Medical. “This milestone represents an exciting new chapter for Tenex technology. We thank our long-standing users for their valuable collaboration and the entire Trice team for their commitment to bringing this innovation to patients and physicians.”
Developed in 2012, the first generation Tenex Health TX System established a minimally invasive ultrasonic approach to treating chronic tendon disease, enabling physicians to intervene early after conservative treatment fails and effectively address the underlying pain.
“The Tenex technology was truly transformative, allowing for safe and effective treatment of multiple challenging musculoskeletal pathologies in a precise and efficient manner,” said Dr. Mederic M. Hall, Sports Medicine Physician at the University of Iowa Health Care. “This 2nd Generation device will allow us to further move the field of ultrasound guided surgery forward with more efficient treatment and expanded applications. Years of user and patient feedback have informed substantive improvements in a device that has already shown significant clinical benefit. This is an exciting time for those of us helping patients with chronic tendinopathy.”
About Trice Medical:
Trice Medical, in Malvern, PA, and Lake Forest, CA, is a leader in minimally invasive medical technologies. The company offers innovative and clinically proven solutions that allow physicians the flexibility to treat in a procedure room, ambulatory surgery center, or hospital setting. Trice Medical’s portfolio includes Seg-WAY™ Endoscopic Release Systems and Tenex® for treating chronic tendinopathy and bony prominences, including diabetic foot ulcers. More information is available at tricemedical.com.
Contacts
Media Contact for Trice Medical:
Lyly Hoang Wight, VP Marketing
(610) 989-8080
marketing@tricemedical.com