CHICAGO (February 16, 2021) – New research published in the February issue of Journal of Wound Care (JWC) concludes the unique micro-anchor skin closure device known as BandGrip is more than five times faster in closing orthopedic incisions compared to traditional sutures. Surgeons have long believed infections can be associated with longer wound closure and overall recovery times. The time-savings finding in anterior cruciate ligament reconstruction (ACLR), simple arthroscopy and general incisions could result in surgeons seeing fewer wound infections following surgery.
“BandGrip allows for more efficient surgical incision closure,” notes the study’s lead author, Brian Cole, M.D., M.B.A., a world-renowned orthopedic sports medicine surgeon, specializing in shoulder, elbow and knee at Midwest Orthopaedics at Rush. “Time to close surgical skin incision is significantly less with the use of BandGrip when compared with conventional sutures.”
For each of the millions of orthopedic surgeries performed every year, a surgical incision closure is required. The medical and financial implications of this part of a surgical procedure is often overlooked yet results in hundreds of dollars in OR time and the potential for infection post-surgery.
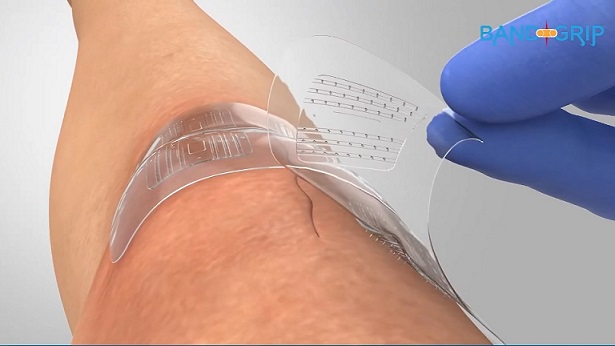
BandGrip is a 3.5”x1.5” bandage that offers a minimally invasive method of skin closure without the use of needles or staples. Its design enables a variety of healthcare professionals to apply and remove the bandage during the management of surgical incisions. BandGrip uses minimally-invasive micro-anchors that gently and securely grip the top layer of skin (dermis) – yet not pierce it – to pull skin edges together.
“We anticipated BandGrip would provide reliable closure with significantly lower time burden when compared with suture closure,” notes Dr. Cole, who consults and is an advisor to BandGrip. “Now, our research confirms it.” Subcuticular needle sutures have long been used by surgeons, but Dr. Cole recognized the potential in the micro-anchor skin closure device when he started using it in the spring of 2019.
As part of the paper, Dr. Cole mentioned another non-invasive skin closure device called Zipline, noting research that shows it is more than three times faster than sutures. Despite the variables between the studies, the greater time-savings found using BandGrip is speaking volumes in the healthcare industry, especially as it relates to the already studied correlation between reduced wound closure times and wound infections. Equally important is the fact that BandGrip is easier to use, more cost-effective and leads to predictable aesthetic outcomes compared to other products that simply report reduced operating room time associated with wound closure.
“We believe further clinical research will show the minimally-invasive makeup of BandGrip is superior to other skin closure devices currently on the market,” says Fred Smith, founder, CEO and Chairman of the Board at BandGrip, a company that holds the same name as its signature product. “BandGrip is the next evolution in wound care. Sutures and staples puncture the skin, which make patients more susceptible to post-surgical complications including inferior cosmetic outcomes at long term follow-up. Further, sutures and staples require removal post-operatively by certified healthcare provider whereas BandGrip can be removed comfortably by the patient in their own homes. With this time-savings study and other evidence-based research on BandGrip, we are strengthening our claim to surgeons that by using our skin closure adhesive – allowing natural healing without foreign materials at or below the skin surface – we can expect fewer complications, wound infections and improved patient and surgeon satisfaction.”
This first study, “Time to closure of orthopaedic surgical incisions: a novel skin closure device versus conventional sutures,” can be found in the February issue of JWC.
Other BandGrip-related research underway and expected to be published this year includes –
- Assessing the incidence of all time and type of wound-related complications following total joint arthroplasty when BandGrip is used verses standard wound closure techniques. The lead researcher is Javad Parvizi, M.D., an orthopedic surgeon based in Philadelphia.
- Comparing BandGrip to a liquid bonding agent for elective knee arthroscopy portal closure to evaluate postoperative outcomes including skin reactions and infections. Particular attention will be made to spitting sutures, which happens on average in 10-to-20% of patients for the lead author, Sherwin S.W. Ho, M.D., a sports medicine specialist for minimally invasive arthroscopic procedures of the shoulder, elbow, hip, knee and ankle in Chicago.
- Investigating the performance of adhesive strips, specifically BandGrip, as cosmetic equivalents to subcuticular sutures. Cosmesis outcomes will be studied by lead researcher Dr. Cole.
“BandGrip is designed to improve the overall quality of surgical incisions,” Mr. Smith adds. “There are many superior benefits to BandGrip – both for patients and their surgeons. We are excited see more clinical research verifying that BandGrip brings to the OR a reduction in complications related to the surgical site in addition to time savings and minimal scarring, proving BandGrip is the most advanced and intuitive skin closure device in the industry.”
For more information on BandGrip’s skin closure technology, visit www.bandgrip.com. See the quick and easy application of BandGrip by clicking here, and the removal of it by clicking here.
About BandGrip, Inc.
Based in Chicago, BandGrip was founded in 2015 with the goal of developing a faster, easier, and less invasive way to safely close wounds. The emerging medical device company canvassed surgeons to identify ways to reduce closure time, improve ambulation, and minimize scarring. As a result, the company’s team of medical professionals developed the patented technology behind the BandGrip Micro-Anchor Skin Closures. Today, BandGrip is advancing the wound-closure market by delivering medical innovations that allow hospitals and doctors to reduce procedure time and improve the patient experience, while envisioning a future where its products can help improve the lives of people around the world.
MEDIA CONTACT INFORMATION
BandGrip, media relations specialist
Karyn Odway, (312) 479-1271