July 12, 2021
LEWISVILLE, Texas–(BUSINESS WIRE)–Orthofix Medical Inc. (NASDAQ:OFIX), a global medical device company with a spine and orthopedics focus, today announced the publication of retrospective data evaluating fusion rates in patients receiving three- and four-level lumbar arthrodesis surgery who were treated adjunctively with the SpinalStim™ bone growth therapy device.
Published in The International Journal of Spine Surgery, the study results show a 92.7-percent fusion rate in patients with and without risk factors such as diabetes, obesity, tobacco use, advanced age and osteoporosis − conditions that have been linked to higher rates of nonunion or bone healing complications.
“We set out to evaluate the success rate of Pulsed Electromagnetic Field Stimulation (PEMF) therapy to demonstrate the efficacy when used as an adjunctive treatment in patients receiving three- and four-level lumbar fusion surgery,” said Dr. Kris Radcliff, Professor of Orthopedic and Neurological Surgery at Thomas Jefferson University. “The fusion rate was very good across all patient types including those who were potentially at high risk of fusion failure. These results reconfirm the benefit PEMF therapy can provide patients when used after multi-level lumbar fusion surgery.”
Following a multicenter retrospective evaluation, data was collected from 55 patients diagnosed with lumbar degenerative disease, spinal stenosis, and/or spondylolistheses who underwent three- or four-level arthrodesis surgery followed by PEMF treatment with the SpinalStim device. At 12-month follow-up, radiographic evaluation of overall fusion rates was 92.7 percent regardless of patient risk factors.
“This data confirms that the efficacy seen in the original Orthofix IDE clinical study of PEMF stimulation with the SpinalStim bone growth therapy system in patients with one- and two-level lumbar procedures is also demonstrated in three- and four-level fusion surgery patients,” said Orthofix President of Global Spine Kevin Kenny. “Importantly, these results include data from patients who were at high clinical risk of fusion failure. It is very rewarding to know that our SpinalStim bone growth therapy system can be a benefit to patients whose recovery from arthrodesis surgery can be challenging.”
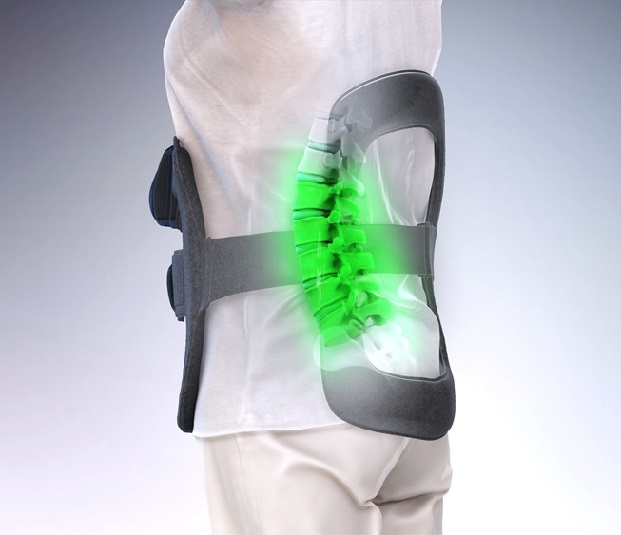
About the SpinalStim Bone Growth Therapy Device
The SpinalStim device is the only bone growth stimulator approved by the U.S. Food and Drug Administration as an adjunct for lumbar spinal fusion procedures and as an option for treating failed spinal fusion surgery. The device emits 360 degrees of PEMF treatment around the fusion site, covering up to five vertebral levels. The SpinalStim system is supported by the STIM onTrack™ mobile app that enables patients to take an active role in their recovery through daily treatment reminders, usage calendars and the sharing of treatment data with physicians.
About Orthofix
Orthofix Medical Inc. is a global medical device company with a spine and orthopedics focus. The Company’s mission is to deliver innovative, quality-driven solutions as we partner with health care professionals to improve patient mobility. Headquartered in Lewisville, Texas, Orthofix’s spine and orthopedics products are distributed in more than 60 countries via the Company’s sales representatives and distributors. For more information, please visit www.Orthofix.com.
Forward-Looking Statements
This communication contains forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended, and Section 27A of the Securities Act of 1933, as amended, relating to our business and financial outlook, which are based on our current beliefs, assumptions, expectations, estimates, forecasts and projections. In some cases, you can identify forward-looking statements by terminology such as “may,” “will,” “should,” “expects,” “plans,” “anticipates,” “believes,” “estimates,” “projects,” “intends,” “predicts,” “potential,” or “continue” or other comparable terminology. These forward-looking statements are not guarantees of our future performance and involve risks, uncertainties, estimates and assumptions that are difficult to predict, including the risks described in Part I, Item 1A under the heading Risk Factors in our Annual Report on Form 10-K for the year ended December 31, 2020 (the “2020 Form 10-K”). In addition to the risks described there, factors that could cause or contribute to such differences may include, but are not limited to: the risk that FDA approvals may be delayed or not be obtained; the risk that surgeons may be slow to adopt the SpinalStim Bone Growth Therapy device; the risk that future patient studies or clinical experience and data may indicate that treatment with the SpinalStim Bone Growth Therapy device does not improve patient outcomes as much as previously believed, or otherwise call into question the benefits of its use to patients, hospitals and surgeons; the risk that the product may not perform as intended and may therefore not achieve commercial success; the risk that competitors may develop superior products or may have a greater market position enabling more successful commercialization; the risk that insurance payers may decline to reimburse healthcare providers for the use of our products.
This list of risks, uncertainties and other factors is not complete. We discuss some of these matters more fully, as well as certain risk factors that could affect our business, financial condition, results of operations, and prospects, in reports we file from time-to-time with the SEC, which are available to read at www.sec.gov. Any or all forward-looking statements that we make may turn out to be wrong (due to inaccurate assumptions that we make or otherwise), and our actual outcomes and results may differ materially from those expressed in these forward-looking statements. You should not place undue reliance on any of these forward-looking statements. Further, any forward-looking statement speaks only as of the date hereof, unless it is specifically otherwise stated to be made as of a different date. We undertake no obligation to update, and expressly disclaim any duty to update, our forward-looking statements, whether as a result of circumstances or events that arise after the date hereof, new information, or otherwise.
Contacts
Alexa Huerta
Investor Relations
Tel 214 937 3190
Denise Landry
Media Relations
Tel 214 937 2529