August 24, 2021
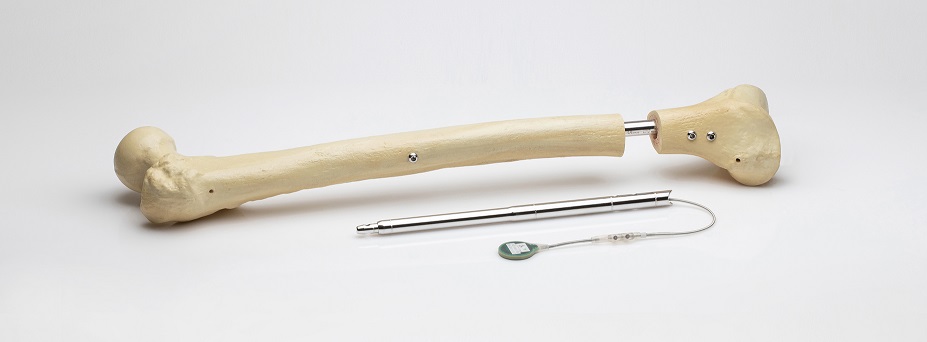
MAHWAH, N.J.–(BUSINESS WIRE)–Stryker has announced the launch of its T2 Alpha Femur Retrograde Nailing System. The new nailing solution features a redefined data-driven nail design,1,2 an advanced locking screw option that provides surgeons with axial stability where desired, and a dedicated proximal targeting system, which may reduce the number of X-ray shots compared to freehand locking.4-7
“Stryker has made significant investments in the T2 Alpha Femur Retrograde Nailing System’s instrumentation platform,” said Eric Tamweber, Vice President and General Manager of Stryker’s Trauma business. “We are excited to now have a complete lower extremity T2 Alpha Nailing platform available for our surgeon customers.”
The T2 Alpha Femur Retrograde Nailing System was created with a new level of innovation, including:
- Data-driven Stryker Orthopaedic Modeling and Analytics (SOMA) designed nails, available in a variety of sizes and radii of curvature, to meet the unique needs of each case and diverse anatomical patient needs.1,2
- Advanced locking screws that are designed to limit relative motion between the nail and screw and provide additional stability when needed.1,3,5
- Guided targeting that is designed to provide more reproducible locking and reduce the number of X-ray shots compared to freehand locking.4,6,7
- Intuitive instrumentation that is inspired by surgeon preferences and designed to improve ease of use, case after case.1
About Stryker
Stryker is one of the world’s leading medical technology companies and, together with its customers, is driven to make healthcare better. The company offers innovative products and services in Orthopaedics, Medical and Surgical and Neurotechnology and Spine that help improve patient and hospital outcomes. More information is available at www.stryker.com.
Follow Stryker’s Trauma business on Instagram, Twitter and LinkedIn.
A surgeon must always rely on his or her own professional clinical judgment when deciding whether to use a particular product when treating a particular patient. Stryker does not dispense medical advice and recommends that surgeons be trained in the use of any particular product before using it in surgery.
The information presented is intended to demonstrate the breadth of Stryker’s product offerings. A surgeon must always refer to the package insert, product label and/or instructions for use before using any of Stryker’s products. Products may not be available in all markets because product availability is subject to the regulatory and/or medical practices in individual markets. Please contact your sales representative if you have questions about the availability of products in your area.
Content ID: T2-LT-8_Rev-1, 08-2021
References:
1. T2 Alpha Retrograde Femur Nailing System Surgical Technique T2-ST-37, 06-2021
2. T2-WP-5_Rev-1, 12-2019
3. Internal Test Report A0030043
4. Adam et al. External distal targeting device for safe insertion of distal locking screws when performing long Gamma Nail: Comparison with the freehand technique. Bone and Joint. Vol 94-B Supp XXXVII. 14. Sept. 2012
5. T2-WP-6_Rev-1, 12-2019
6. M.Ehlinger et al. Distal targeting device for long Gamma nail®. Monocentric observational study. Orthopaedics and Traumatology:Surgery and Research. Vol 99-Issue 7. November 2013, Pages 799-804
7. T2-WP-8, 03-2019
Contacts
Media Contacts
Andrea Sampson, Sampson Public Relations Group
asampson@sampsonprgroup.com
562.304.0301