Modena, September 27, 2021 / OrthoSpineNews
Creativity is an essential fuel for the medical device industry, also when it comes to commercial product names. Through its commercial product names, Italy based Tsunami Medical has praised its heritage by conveying a sense of belonging and tribute to the country’s many beautiful islands. The company’s “wave of innovation” is metaphorically reflected in these incredible features of nature.
Since its very first product launches of latest innovations in 3D printed spinal implant technology back in 2014, Tsunami Medical has used many Italian islands’ names for the branding of its products.
After acknowledging that two of its product commercial names (Capri and Panarea) are also present in the catalogue of Stryker Corporation for Spinal Implants (although for different intended uses), Tsunami Medical is actively intervening in the re-branding of these products.
Tsunami Medical appreciates to not contribute to any kind of confusion towards its well respected customers and the other company involved.
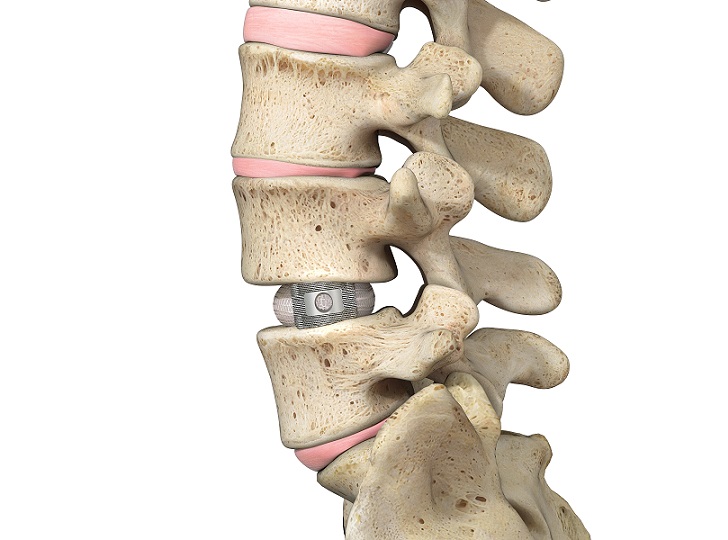
Thus, Capraia and Pianosa will represent the new commercial names of their ACIF and PLIF Interbody Fusion cages respectively and Tsunami Medical will stop actively promoting the former commercial names with immediate effect.
Further details will be communicated to Tsunami Medical’s customers directly.
About the Company
Tsunami Medical was founded in 1997, manufacturing invasive diagnostic devices as subcontractor of big manufacturing companies. Over the years the Company has bought the Bloodline trademark, very well known in the international biopsy and vertebroplasty spinal markets.
In 2010 Tsunami Medical started the design and manufacturing of Selective Laser Melting (SLM) technology, resulting in the currently available implant product portfolio.
Located in the heart of the “Biomedical Valley”, nearby Modena (Italy), the manufacturing facility extends on 1200 square meters with modern machinery and clean rooms class ISO 8, 13485 certified to design and manufacture medical devices.