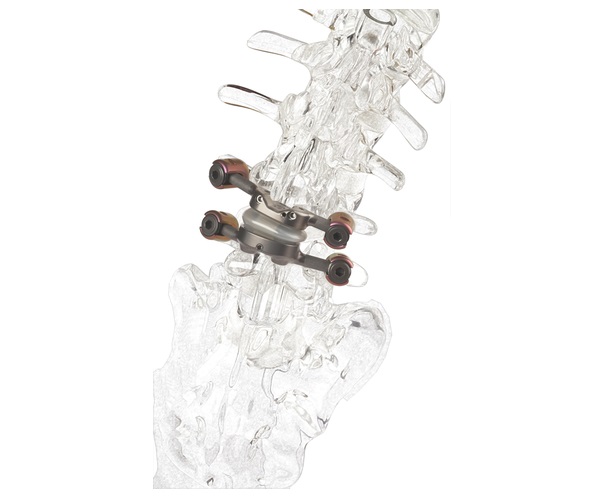
CARLSBAD, Calif., Sept. 06, 2022 (GLOBE NEWSWIRE) — SeaSpine Holdings Corporation (NASDAQ: SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, today announced the full commercial launch of the WaveForm TA (TLIF Articulating) Interbody System.
The WaveForm TA Interbody System is designed to deliver an interbody to the anterior portion of the disc space to optimize sagittal alignment and endplate support. With an array of footprint and lordotic options from which to choose, surgeons have the ability to address specific anatomical needs intraoperatively and without adversely impacting surgical workflow.
“WaveForm TA is an exciting addition to our interbody portfolio and provides a best-in-class offering in what we estimate is the $175 million U.S. TLIF market for 3D-printed and solid titanium products,” stated Dennis Cirino, Senior Vice President, Global Spinal Systems. “This system provides a versatile and reproducible lumbar interbody solution, while providing efficiency through cross-compatible instrumentation. This allows surgeons the flexibility of choosing from multiple SeaSpine implant families with only one set of familiar instrumentation.”
WaveForm interbody devices offer the next level of 3D-printed architectural innovation, balancing key geometric and manufacturing advancements without compromising clinical requirements. The WaveForm TA Interbody System utilizes innovative WaveForm technology to deliver a highly porous, yet robust, architecture intended to optimize subsidence resistance, implant stiffness, and orthobiologics packability, while maintaining radiographic visualization during intraoperative and postoperative imaging.
“The WaveForm TA implant has a very thoughtful design. The porosity allows for bony in-growth while also balancing the ideal roughness,” said Dr. Alex Mohit of NeoSpine and Tacoma General Hospital. “The simple and efficient instrumentation has been fine-tuned to insert the implant safely and accurately. I have been extremely impressed with the results.”
About SeaSpine
SeaSpine (www.seaspine.com) is a global medical technology company focused on the design, development, and commercialization of surgical solutions for the treatment of patients suffering from spinal disorders. SeaSpine’s complete procedural solutions feature its market-leading FLASH™ Navigation, a system designed to improve accuracy of screw placement and provide a cost-effective, rapid, radiation-free solution to surgical navigation, and a comprehensive portfolio of spinal implants and orthobiologics to meet the varying combinations of products that neurosurgeons and orthopedic spine surgeons need to facilitate spinal fusion in degenerative, minimally invasive surgery (MIS), and complex spinal deformity procedures on the lumbar, thoracic and cervical spine. With product development expertise in advanced optics, software, orthobiologic sciences and spinal implants, SeaSpine can offer its surgeon customers a complete solution to meet their patients’ evolving clinical needs. SeaSpine currently markets its products in the United States and in approximately 30 countries worldwide.
Forward-Looking Statements
SeaSpine cautions you that statements included in this news release that are not a description of historical facts are forward-looking statements that are based on the Company’s current expectations and assumptions. Such forward-looking statements include, but are not limited to, statements relating to: the design of WaveForm TA and its ability to provide a versatile lumbar interbody solution for surgeons; the ability of WaveForm TA to balance key geometric and manufacturing advancements without compromising clinical utility; the ability of WaveForm technology to deliver a highly porous and robust interbody solution and appropriately balance subsidence resistance, implant stiffness, and orthobiologics packability, while maintaining radiographic visualization during intraoperative and postoperative imaging. Among the factors that could cause or contribute to material differences between the Company’s actual results and the expectations indicated by the forward-looking statements are risks and uncertainties that include, but are not limited to: unexpected expense and delay, including as a result of developing and supporting the launch of new products, such as WaveForm TA, the fact that newly launched products may require substantial additional development activities, which could introduce further expense and delay, or as a result of delay in timely obtaining regulatory clearances; the ability of newly launched products, such as WaveForm TA, to perform as designed and intended and to meet the needs of surgeons and patients, including as a result of the lack of substantial clinical validation of products following limited commercial (or “alpha”) launch; and other risks and uncertainties more fully described in the Company’s news releases and periodic filings with the Securities and Exchange Commission. The Company’s public filings with the Securities and Exchange Commission are available at www.sec.gov. You are cautioned not to place undue reliance on forward-looking statements, which speak only as of the date when made. SeaSpine does not intend to revise or update any forward-looking statement set forth in this news release to reflect events or circumstances arising after the date hereof, except as may be required by law.
Investor Relations Contact
Leigh Salvo
(415) 937-5402
ir@seaspine.com
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/9f8115f4-dc75-4446-9d4f-83adcc7669ae