Milestone aligns with ten-year anniversary of Mobi-C FDA approval for treatment of one and two levels of the cervical spine
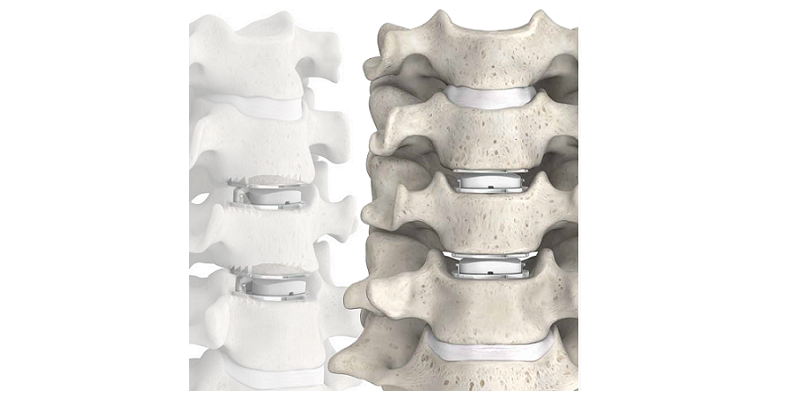
WESTMINSTER, Colorado, January 30, 2023 /OrthoSpineNews/ – ZimVie Inc. (Nasdaq: ZIMV), a global life sciences leader in the dental and spine markets, today announced that over 200,000 Mobi-C Cervical Discs have been implanted worldwide. This includes patients treated in over 25 countries since the first surgery was completed with the device in France in 2004. In 2013, Mobi-C became the first cervical disc to win approval from the U.S. Food and Drug Administration for the treatment of more than one level of the cervical spine.
The FDA has determined Mobi-C to be statistically superior to fusion at 7 years for two-level cervical disc replacement, based on the primary study endpoint of a prospective, concurrently controlled and randomized, multi-center clinical trial.1 At 10 years, all patient-reported outcomes were equivalent to or improved from 7 years.2
“Mobi-C is differentiated by substantial, long-term clinical data and we are proud to announce this milestone, reflecting tens of thousands of patient lives restored with our novel device,” noted Rebecca Whitney, Global President of ZimVie Spine. “As we mark the ten-year anniversary of Mobi-C FDA approval for treatment of one and two levels of the cervical spine, we are excited to embrace 2023 as ‘the Year of Mobi-C’ by acknowledging and celebrating throughout the year the clinical success our surgeon customers have realized for their patients.”
Dr. Armen Khachatryan, board-certified orthopedic surgeon at The Disc Replacement Center in West Jordan, Utah, added, “Having participated in the Mobi-C Investigational Device Exemption study over a decade ago, I am encouraged that disc replacement has become an emerging standard of care for patients with cervical disc degeneration. ZimVie’s dedication to this space has enabled so many patients to benefit from the technology. It is fitting to acknowledge this significant milestone for Mobi-C and the advancement of cervical disc arthroplasty. I sincerely hope that this transition from fusion to motion preserving disc replacement technology will not only continue but accelerate in the coming years.”
Common post-operative risks from surgery with the Mobi-C include pain in the neck, arm, back, shoulder, or head, and dysphagia. For complete indications, contraindications, warnings, and risks on the Mobi-C Cervical Disc or to find more information on other ZimVie Spine solutions, please visit https://www.zimvie.com/en/spine.html.
About the Mobi-C Cervical Disc
Mobi-C is the first cervical disc prosthesis approved by the FDA for reconstruction of a cervical disc at both one and two levels (C3-C7). Mobi-C is a cobalt chromium alloy and polyethylene mobile-bearing prosthesis that is inserted in a single step, without requiring bone chiseling or other vertebral anchorage such as screws or keels. The Mobi-C Cervical Disc Prosthesis is indicated in skeletally mature patients for reconstruction of the disc from C3-C7 following discectomy at one level or two contiguous levels for intractable radiculopathy (arm pain and/or neurological deficit) with or without neck pain or myelopathy due to abnormality localized to the level of the disc space and at least one of the following conditions confirmed by radiographic imaging (CT, MRI or X-rays): herniated nucleus pulposus, spondylosis (defined by the presence of osteophytes) and/or visible loss of disc height compared to adjacent levels. The Mobi-C Cervical Disc Prosthesis is implanted using an anterior approach. Patients should have failed at least six weeks of conservative treatment or demonstrated progressive signs or symptoms despite nonoperative treatment prior to implantation of the Mobi-C Cervical Disc Prosthesis.
About ZimVie
ZimVie is a global life sciences leader in the dental and spine markets that develops, manufactures, and delivers a comprehensive portfolio of products and solutions designed to treat a wide range of spine pathologies and support dental tooth replacement and restoration procedures. The company was founded in March 2022 as an independent, publicly traded spin-off of the Dental and Spine business units of Zimmer Biomet to breathe new life, dedicated energy, and strategic focus to its portfolio of trusted brands and products. From its headquarters in Westminster, Colorado, and additional facilities around the globe, the company serves customers in over 70 countries worldwide with a robust offering of dental and spine solutions including differentiated product platforms supported by extensive clinical evidence. For more information about ZimVie, please visit us at www.ZimVie.com. Follow @ZimVie on Twitter, Facebook, LinkedIn, or Instagram.
Cautionary Note Regarding Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. Forward-looking statements include, but are not limited to, statements concerning ZimVie’s expectations, plans, prospects, and product and service offerings, including new product launches and potential clinical successes. Such statements are based upon the current beliefs, expectations, and assumptions of management and are subject to significant risks, uncertainties, and changes in circumstances that could cause actual outcomes and results to differ materially from the forward-looking statements. For a list and description of some of such risks and uncertainties, see ZimVie’s periodic reports filed with the U.S. Securities and Exchange Commission (SEC). These factors should not be construed as exhaustive and should be read in conjunction with the other cautionary statements that are included in ZimVie’s filings with the SEC. Forward-looking statements speak only as of the date they are made, and ZimVie disclaims any intention or obligation to update or revise any forward-looking statements, whether as a result of new information, future events, or otherwise. Readers of this press release are cautioned not to rely on these forward-looking statements, since there can be no assurance that these forward-looking statements will prove to be accurate. This cautionary note is applicable to all forward-looking statements contained in this press release.
Media Contact Information:
ZimVie
Laura Driscoll • Laura.Driscoll@ZimVie.com
(774) 284-1606
ZimVie Spine
Mark Richards • Mark.Richards@ZimVie.com
(512) 913-9572
Investor Contact Information:
Gilmartin Group LLC
Marissa Bych • Marissa@gilmartinir.com
References:
1 Radcliff K, Davis RJ, Hisey MS, et al. Long-term evaluation of cervical disc arthroplasty with the Mobi-C Cervical Disc: a randomized, prospective, multicenter clinical trial with seven-year follow-up. Int J Spine Surg 2017;11(4):244-262. DOI: https://doi.org/10.14444/4031
2 Kim K, Hoffman G, Bae H, et al. Ten-Year Outcomes of 1- and 2-Level Cervical Disc Arthroplasty From the Mobi-C Investigational Device Exemption Clinical Trial. Neurosurgery. 2021;88(3):497-505. DOI: https://doi.org/10.1093/neuros/nyaa459