Further real-world evidence supporting the efficacy and use of ReActiv8® Restorative NeurostimulationTM for the treatment of intractable Chronic Low Back Pain
DUBLIN–July 20, 2023 (BUSINESS WIRE)–Mainstay Medical Holdings plc today announced the publication of three-year clinical data from the Post-Market Clinical Follow-up (PMCF) study of 33 ReActiv8 patients from five medical centres across the United Kingdom. The three-year results, published in British Journal of Pain, demonstrated that a substantial portion of patients experienced statistically significant improvements in measures of pain (NRS), disability (ODI) and quality of life (EQ-5D-5L). The publication can be found here: https://journals.sagepub.com/doi/10.1177/20494637231181498.
These results indicate that the response to ReActiv8 for these patients is durable and improves over time, validating ReActiv8’s restorative mechanism of action. In addition, these real-world outcomes are consistent with the three-year data from the pivotal ReActiv8-B clinical trial, announced in September 2022, as shown in the following table:
| % of Patients Showing Clinical Improvements at 3 Years | ||||||||
| Outcome Measures | ReActiv8-B (N=133) | PMCF (N=33) | ||||||
| CLBP Remitter (VAS ≤ 2.5/NRS ≤3) | 64% | 70% | ||||||
| ≥ 50% reduction in low back pain | 77% | 67% | ||||||
| ≥ 15-point reduction in ODI | 71% | 55% | ||||||
| ≥50% in VAS/NRS and/or ≥20 points in ODI | 79% | 75% | ||||||
| Completion Rate | 76% | 79% |
Dr. Simon Thomson MBBS FFPMRCA, Consultant Lead at the Pain and Neuromodulation Centre, Mid and South Essex University Hospitals NHS, Essex, UK, stated: “These results demonstrate durability and safety of this therapy in chronic back pain sufferers who would have continued to be crippled and dominated by their symptoms but for ReActiv8. These patients, drawn from usual UK Pain clinics, are now as good as those seen in the continuation cohort from the more highly selected randomised ReActiv8-B trial.”
Jason Hannon, CEO of Mainstay Medical, stated: “We are pleased to add these compelling results to the growing global body of positive peer-reviewed evidence supporting the ability of ReActiv8 to provide positive long-term outcomes to this severely affected patient population. Most importantly, the continued improvement in patient outcomes observed in the real-world setting is consistent with the results from our controlled clinical trials.”
About ReActiv8®
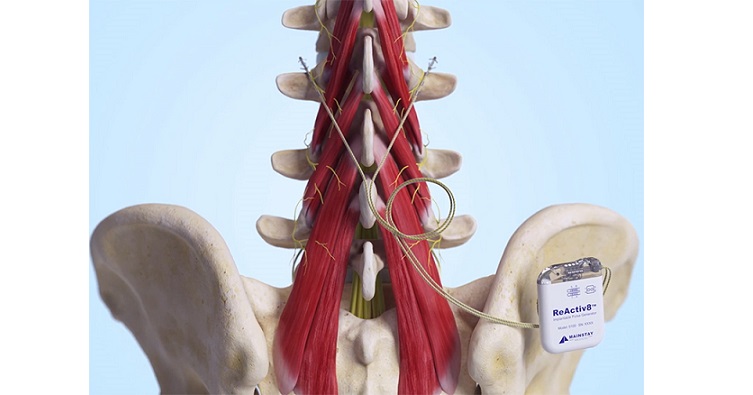
ReActiv8 is an implantable medical device designed to treat adults with intractable chronic low back pain (CLBP) associated with multifidus muscle dysfunction, which may be evidenced by imaging or physiological testing. Candidates for ReActiv8 are patients with multifidus muscle dysfunction who have failed other forms of therapy (including pain medication and physical therapy) and are not candidates for spine surgery. ReActiv8 has received regulatory approval in several geographic areas, and is commercially available in the European Economic Area, Australia, the UK, and the US.
About Mainstay Medical
Mainstay Medical is a medical device company focused on commercializing its innovative implantable Restorative Neurostimulation system, ReActiv8®, for people with disabling mechanical CLBP. Mainstay Medical is headquartered in Dublin, Ireland and has subsidiaries operating in Ireland, the United States, Australia, Germany, and the Netherlands.
Further information can be found at www.mainstaymedical.com.
Forward-Looking Statements
All statements in this announcement other than statements of historical fact are, or may be deemed to be, forward-looking statements. These forward-looking statements may include, without limitation, statements regarding the company’s intentions, beliefs or current expectations concerning, among other things, the company’s research studies and results, commercial efforts and performance, financial position, financing strategies, product design and development, regulatory applications and approvals, and reimbursement arrangements.
Forward-looking statements involve risk and uncertainty and are not guarantees of future performance. Actual results may differ materially from those described in, or suggested by, the forward-looking statements. A number of factors could cause results and developments to differ materially from those expressed or implied by the forward-looking statements herein, including, without limitation, the risks and uncertainties included in the company’s Annual Report for the year ended 31 December 2022, which should be read in conjunction with the company’s public disclosures (available on the company’s website (www.mainstaymedical.com)). The forward-looking statements herein speak only as of the date of this announcement.
Contacts
PR and IR Enquiries:
LifeSci Advisors, LLC
Brian Ritchie
Tel: + 1 (212) 915-2578
Email: britchie@lifesciadvisors.com
FTI Consulting (for Ireland)
Jonathan Neilan or Patrick Berkery
Tel. : +353 1 765 0886
Email: mainstay@fticonsulting.com
Mainstay Medical
Corporate Communications
Email: Media@mainstaymedical.com