Reverse HRS Case Study, Stephen J. Zabinski, MD
December 27, 2023 – OrthoSpineNews –
Summary
The patient is JD, a 64-year-old male who presented with 2-3 years of chronic hip pain. He had a history of previous physical therapy and chiropractic care with no relief. He also underwent one unsuccessful hyaluronic acid injection treatment.
We initially met JD in March 2023 and upon examination he presented with severe loss of motion in the hip and required the use of a walker. Also, one leg was half an inch shorter than the other leg. X-rays revealed advanced “bone-on-bone” arthritis with bones spurs surrounding the joint.
At this time, we discussed the pros and cons of hip replacement as his next best option. We described current best practices and outlined the process for traditional hip implantation. Next, we discussed his potential candidacy to enroll in a clinical trial using the Reverse Hip Replacement System (Reverse HRS). We reviewed scale models of traditional hip replacement devices and the Reverse HRS. I explained how the ball and socket design of the traditional implant can dislocate with extremes of rotation. I showed him the stability of the Reverse HRS in extremes of rotation.
We reviewed the growing database of research and experience with the Reverse HRS. And we discussed the pros and cons of study participation. He was very interested and motivated to proceed with enrollment into the Reverse HRS trial.
Study Overview
In January 2023, Hip Innovation Technology initiated their FDA (Food and Drug Administration) Investigational Device Exemption (IDE) approved pivotal clinical study to further evaluate the company’s Reverse Hip Replacement System (Reverse HRS) for use in Primary Total Hip Arthroplasty (THA).
The study entitled, “Safety and Effectiveness of the Hip Innovation Technology Reverse Hip Replacement System in Primary Total Hip Arthroplasty. A Pivotal Study,” is designed to evaluate the safety and effectiveness of the Reverse HRS in patients undergoing Total Hip Replacement. Safety will be assessed through the collection of device-related adverse events and patient quality of life metrics.
Effectiveness will be evaluated using clinical, radiologic, and patient-reported outcomes.
Orthopedic surgeons interested in participating in this study, or would like more information, please contact Hip Innovation Technology here: CorporateDevelopment@HIT-IRH.com.
Results
JD was enrolled in the study, and, after randomization he was selected to receive the Reverse HRS. The surgical procedure took place without incident on March 28, 2023. The Reverse HRS was implanted with an anterior approach. Upon immediate post-operative evaluation, his x-rays were normal and his leg lengths were restored back to normal. He was released and returned home the same day as his operation.
At his first post-op follow-up on April 11, 2023, he did not require a walker and was using a cane. His x-rays were normal and his wound was healing well. At this time, he was prescribed to engage in an ongoing walking exercise program. Upon his next post-op evaluation on May 12, 2023, he presented with no issues, his wound was healing well and he displayed excellent motion and strength. He was prescribed to continue with exercises.
On September 25, 2023, we met with JD for his six-month check-up. Aside from occasional pain, he was feeling well, and showed excellent motion, full strength, and no instability. He was advised to continue walking and add strengthening exercises.
(sidebar)
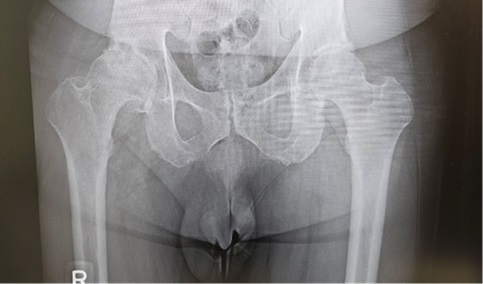
Before hip replacement surgery
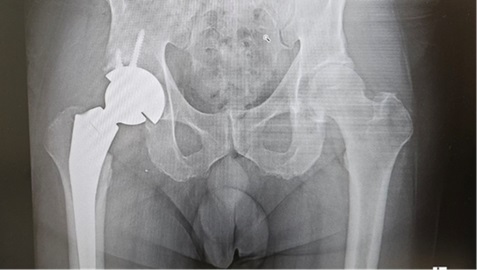
After implant with Reverse HRS
Lessons Learned
As we proceed through this landmark clinical trial, we are discovering extremely valuable insights along the way. The biggest lesson is the vital importance of correctly setting the leg length and the tension of the soft tissues. When this is done, the implant delivers incredible stability. In fact, it was actually hard to get the hip to pop out of place when we tried it in the OR. I actually had to use a small bone hook that I put around the trial implant to pull the leg down and out to the side to get it to dislocate.
The other aspect of the implant that we learned is that the femoral stem is very stout and slightly larger than traditional femoral stems. It is a strong implant. Occasionally, people with a tight bone canal will require you to ream the canal so the implant will fit.
Insights – Pelvic Spinal Disorder
Finally, based on our experience with metal-on-metal implants, the issue of spinal pelvic disorder and the increasing occurrence of instability and dislocation in these patients seems to be mitigated. Based on our experience, the Reverse HRS should eliminate this issue. It is superior to anything we have on the market today. Current implants still have the larger ball, or dual mobility ball for stability, and at extremes of rotation, if the pelvis tilts, the ball is going to pop out of place. With the Reverse HRS when you get to extremes of rotation the femoral cup actually becomes further behind the acetabular ball and becomes more locked in place.
Corresponding Author
Stephen Zabinski, MD, Director of the Division of Orthopaedic Surgery, Shore Medical Center in Ocean City, New Jersey
Email: sjzabinski@comcast.net
Acknowledgements
Hip Innovation Technology (http://www.hipinnovationtechnology.com/index.html)
IRB — Nor Consult, LLC (www.nor-consult.com)
The team at Shore Ortho University Associates (https://www.shoreorthopaedic.com/)
The operating room team at Shore Medical Center (https://shoremedicalcenter.org/)








