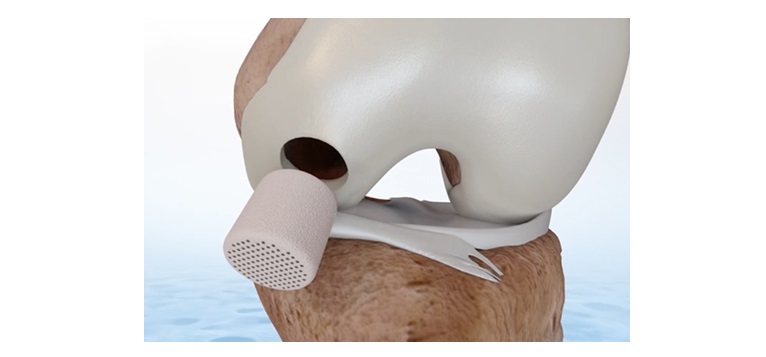
10 January 2024 – Smith+Nephew (LSE:SN, NYSE:SNN), the global medical technology company, announces that it has completed the acquisition of CartiHeal, developer of Agili-C, a novel sports medicine technology for cartilage regeneration in the knee.
Agili-C is an off-the-shelf one-step treatment for osteochondral (bone and cartilage) lesions with a broader indication than existing treatments. It is indicated to treat a wide patient population, including those with lesions in knees with mild to moderate osteoarthritis, a previously unaddressed condition, as well as the approximately 700,000 patients1 that receive cartilage repair annually in the US.
“With its proven superiority2 to current standard of care, Agili-C has the potential to transform cartilage repair outcomes,” said Scott Schaffner, President Sports Medicine, Smith+Nephew. “Our expertise in regenerative therapy and leadership in knee repair gives me great confidence that this will be a significant value creator for Smith+Nephew.”
Smith+Nephew paid $180 million on completion, with up to a further $150 million contingent on future financial performance, as previously disclosed.
Enquiries
| Investors / Analysts Andrew Swift Smith+Nephew | +44 (0) 1923 477433 |
| Media Charles Reynolds Smith+Nephew | +44 (0) 1923 477314 |
| Susan Gilchrist / Ayesha Bharmal Brunswick | +44 (0) 20 7404 5959 |
About Smith+Nephew
Smith+Nephew is a portfolio medical technology business that exists to restore people’s bodies and their self-belief by using technology to take the limits off living. We call this purpose ‘Life Unlimited’. Our 19,000 employees deliver this mission every day, making a difference to patients’ lives through the excellence of our product portfolio, and the invention and application of new technologies across our three global business units of Orthopaedics, Advanced Wound Management and Sports Medicine & ENT.
Founded in Hull, UK, in 1856, we now operate in more than 100 countries, and generated annual sales of $5.2 billion in 2022. Smith+Nephew is a constituent of the FTSE100 (LSE:SN, NYSE:SNN). The terms ‘Group’ and ‘Smith+Nephew’ are used to refer to Smith & Nephew plc and its consolidated subsidiaries, unless the context requires otherwise.
For more information about Smith+Nephew, please visit www.smith-nephew.com and follow us on X, LinkedIn, Instagram or Facebook.
Forward-looking Statements
This document may contain forward-looking statements that may or may not prove accurate. For example, statements regarding expected revenue growth and trading profit margins, market trends and our product pipeline are forward-looking statements. Phrases such as “aim”, “plan”, “intend”, “anticipate”, “well-placed”, “believe”, “estimate”, “expect”, “target”, “consider” and similar expressions are generally intended to identify forward-looking statements. Forward-looking statements involve known and unknown risks, uncertainties and other important factors that could cause actual results to differ materially from what is expressed or implied by the statements. For Smith+Nephew, these factors include: risks related to the impact of Covid, such as the depth and longevity of its impact, government actions and other restrictive measures taken in response, material delays and cancellations of elective procedures, reduced procedure capacity at medical facilities, restricted access for sales representatives to medical facilities, or our ability to execute business continuity plans as a result of Covid; economic and financial conditions in the markets we serve, especially those affecting healthcare providers, payers and customers (including, without limitation, as a result of Covid); price levels for established and innovative medical devices; developments in medical technology; regulatory approvals, reimbursement decisions or other government actions; product defects or recalls or other problems with quality management systems or failure to comply with related regulations; litigation relating to patent or other claims; legal and financial compliance risks and related investigative, remedial or enforcement actions; disruption to our supply chain or operations or those of our suppliers (including, without limitation, as a result of Covid); competition for qualified personnel; strategic actions, including acquisitions and disposals, our success in performing due diligence, valuing and integrating acquired businesses; disruption that may result from transactions or other changes we make in our business plans or organisation to adapt to market developments; relationships with healthcare professionals; reliance on information technology and cybersecurity; disruptions due to natural disasters, weather and climate change related events; changes in customer and other stakeholder sustainability expectations; changes in taxation regulations; effects of foreign exchange volatility; and numerous other matters that affect us or our markets, including those of a political, economic, business, competitive or reputational nature. Please refer to the documents that Smith+Nephew has filed with the U.S. Securities and Exchange Commission under the U.S. Securities Exchange Act of 1934, as amended, including Smith+Nephew’s most recent annual report on Form 20-F, which is available on the SEC’s website at www. sec.gov, for a discussion of certain of these factors. Any forward-looking statement is based on information available to Smith+Nephew as of the date of the statement. All written or oral forward-looking statements attributable to Smith+Nephew are qualified by this caution. Smith+Nephew does not undertake any obligation to update or revise any forward-looking statement to reflect any change in circumstances or in Smith+Nephew’s expectations.
◊ Trademark of Smith+Nephew. Certain marks registered in US Patent and Trademark Office.
References
- Medtech 360, Sports Medicine Devices, Market Analysis, US, 2018, Millennium Research Group, Inc.
- The U.S. Food and Drug Administration (FDA) granted Agili-C Breakthrough Device designation status in 2020 and Premarket Approval (PMA) in March 2022. PMA approval was granted based on the results of a two-year randomised controlled trial (N=251) that confirmed superiority of Agili-C over current standard of care – microfracture and debridement for the treatment of knee joint surface lesions, chondral and osteochondral defects. Study inclusion criteria included patients with mild and moderate osteoarthritis.