Company is initiating its pivotal multicenter, prospective, randomized CLARITY trial to build on clinical evidence demonstrating superior surgical outcomes when Nociscan is utilized in surgical decisioning
5 additional Key Opinion Leader (KOL) surgeons expected to adopt Nociscan through the Alphatec strategic partnership in 1H24
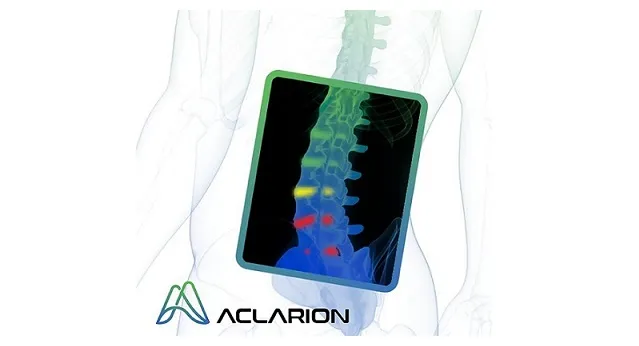
BROOMFIELD, CO, Feb. 05, 2024 (GLOBE NEWSWIRE) — via NewMediaWire — Aclarion, Inc., (“Aclarion” or the “Company”) (Nasdaq: ACON, ACONW), a healthcare technology company that is leveraging biomarkers and proprietary augmented intelligence algorithms to help physicians identify the location of chronic low back pain, announced today that it has completed several recent financing transactions that have significantly reduced debt and increased available cash. The Company believes these financing transactions will assist in achieving key milestones that include initiating the CLARITY trial, onboarding additional KOL surgeons through the strategic partnership with Alphatec, and continuing to increase Nociscan orders.
The financing transactions include raising just under $3M of gross proceeds under the previously disclosed $10M equity line and reducing the Company’s outstanding non-convertible debt by $1.5M through the conversion of debt into equity. The Company believes these recent financing transactions were on terms more favorable than those available in the public capital markets.
With these proceeds, Aclarion will initiate CLARITY, a prospective, multicenter, randomized clinical trial designed to validate the superior surgical outcomes that have been shown to result when surgical interventions are targeted against intervertebral discs identified to be painful by Nociscan technology. CLARITY is expected to enroll up to 300 patients across approximately 8 sites with Nicholas Theodore, MD, FACS, FAANS, Director of the Neurosurgical Spine Center and Professor of Neurosurgery, Orthopedics, and Biomedical Engineering at Johns Hopkins, serving as principal investigator.
Ryan Bond, Chief Strategy Officer at Aclarion, said, “We have made great strides in advancing the clinical data supporting the utilization of Nociscan as a tool to improve surgical decisioning in spine surgery. CLARITY represents the pivotal study we believe will lead to national coverage decisions from every payer of significance, should CLARITY results confirm those from existing peer reviewed published data. Since Nociscan is a diagnostic, we expect to quickly and cost effectively demonstrate the value of our technology in comparison to the costly and lengthy clinical trials typical for implantable devices.”
The Company is also advancing through the first phases of the Alphatec strategic partnership. Aclarion and Alphatec leadership are in the process of identifying 5 KOL surgeons who are currently using Alphatec’s AlphaInformatiX platform and want to add Nociscan data to their preprocedural planning and surgical decisioning process. The Company expects to add 5 new KOLs to the Nociscan network in the first half of 2024.
“As we continue to onboard imaging centers to meet the needs of our core panel of 10 KOLs, we are excited to work with Alphatec to add an additional 5 KOLs to the Aclarion network. Due to the large number of surgeons driving Alphatec growth, we have the luxury of building our new KOL panel with surgeons who already have access to an imaging center capable of performing Nociscans. This will greatly accelerate our ability to reap the benefits of surgeon feedback and accelerate not only Nociscan utilization but progression through the sequential steps of the strategic partnership,” said Brent Ness, Aclarion CEO.
The Company will provide updates as clinical sites are added to CLARITY and KOLs are onboarded through the strategic partnership.
For information about Nociscan please contact Aclarion at info@aclarion.com.
About Aclarion, Inc.
Aclarion is a healthcare technology company that leverages Magnetic Resonance Spectroscopy (“MRS”), proprietary signal processing techniques, biomarkers, and augmented intelligence algorithms to optimize clinical treatments. The Company is first addressing the chronic low back pain market with Nociscan, the first, evidence-supported, SaaS platform to noninvasively help physicians distinguish between painful and nonpainful discs in the lumbar spine. Through a cloud connection, Nociscan receives magnetic resonance spectroscopy (MRS) data from an MRI machine for each lumbar disc being evaluated. In the cloud, proprietary signal processing techniques extract and quantify chemical biomarkers demonstrated to be associated with disc pain. Biomarker data is entered into proprietary algorithms to indicate if a disc may be a source of pain. When used with other diagnostic tools, Nociscan provides critical insights into the location of a patient’s low back pain, giving physicians clarity to optimize treatment strategies. For more information, please visit www.aclarion.com.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934 about the Company’s current expectations about future results, performance, prospects and opportunities. Statements that are not historical facts, such as “anticipates,” “believes” and “expects” or similar expressions, are forward-looking statements.
This press release contains forward-looking statements, among other items, regarding the Company’s capital raising plans and needs for additional capital to continue fund ongoing operations. Our present plans to raise additional capital may change, and our plans will be subject to market conditions and other factors. All of our forward-looking statements are based on the current plans and expectations of management and are subject to a number of uncertainties and risks that could significantly affect the Company’s current plans and expectations, as well as future results of operations and financial condition. These and other risks and uncertainties are discussed more fully in our filings with the Securities and Exchange Commission. Readers are encouraged to review the section titled “Risk Factors” in the Company’s amended Annual Report on Form 10-K, as filed with the Securities and Exchange Commission on June 12, 2023, as well as other disclosures contained in such Annual Report and subsequent filings made with the Securities and Exchange Commission. Forward-looking statements contained in this announcement are made as of this date and the Company undertakes no obligation to publicly update or revise any forward-looking statements, whether as a result of new information, future events or otherwise.
Investor Contacts:
Kirin M. Smith
PCG Advisory, Inc.
646.823.8656
ksmith@pcgadvisory.com
Media Contacts:
Jodi Lamberti
SPRIG Consulting
612.812.7477
jodi@sprigconsulting.com