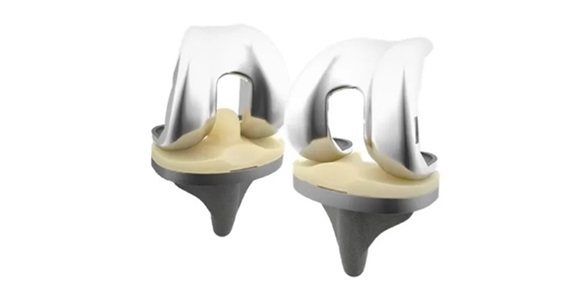
WARSAW, Ind., and ZURICH, Nov. 18, 2024 /PRNewswire/ — Zimmer Biomet Holdings, Inc. (NYSE: ZBH and SIX: ZBH), a global medical technology leader, today received the CE Mark for its Persona® Revision Knee System. This addition to the Persona family builds on Zimmer Biomet’s commitment to personalized solutions, providing surgeons with advanced tools for revision knee arthroplasty.
“After the successful introduction of Persona Revision Knee in the United States and other markets, the CE Mark certification and commercial launch extends its availability significantly so that more surgeons have access to an innovative solution that can enhance patient outcomes,” said Joe Urban, President, Knees at Zimmer Biomet.
The Persona Revision Knee offers a personalized fit and optimized function for patients undergoing revision knee surgery. The modular system allows for intraoperative flexibility, so surgeons can address a wide range of complex cases with precision and ease. The system features Zimmer Biomet’s proprietary Trabecular Metal™ Technology, which promotes long-term bone in-growth and improves implant fixation.1 2 3
“We are excited to introduce the Persona Revision Knee to expand our industry-leading knee portfolio and to provide our customers and their patients with a system that is truly personalized, adaptable, and designed to restore natural knee motion,” said Wilfred van Zuilen, Group President EMEA, Zimmer Biomet.
The Persona Revision Knee system is the leading product in the over $700 million U.S. knee revision market.4 The system’s anatomic components and intuitive instrumentation streamline the revision surgery process, reducing complexity while improving precision.5 6 7 8
Surgeons can make intraoperative adjustments to achieve optimal alignment and fit for each patient, which can lead to improved stability, mobility, and implant longevity.
This launch supports Zimmer Biomet’s Mission to alleviate pain and improve the quality of life for patients worldwide through innovative and clinically proven solutions.
Persona Revision Knee
For more information on the Persona Revision Knee and Zimmer Biomet’s comprehensive portfolio of orthopedic products, visit https://www.zimmerbiomet.com/en/products-and-solutions/specialties/knee/persona-revision.html
About Zimmer Biomet
Zimmer Biomet is a global medical technology leader with a comprehensive portfolio designed to maximize mobility and improve health. We seamlessly transform the patient experience through our innovative products and suite of integrated digital and robotic technologies that leverage data, data analytics and artificial intelligence.
With 90+ years of trusted leadership and proven expertise, Zimmer Biomet is positioned to deliver the highest quality solutions to patients and providers. Our legacy continues to come to life today through our progressive culture of evolution and innovation.
For more information about our product portfolio, our operations in 25+ countries and sales in 100+ countries or about joining our team, visit www.zimmerbiomet.com or follow on LinkedIn at www.linkedin.com/company/zimmerbiomet or X / Twitter at www.twitter.com/zimmerbiomet.
Cautionary Statement Regarding Forward-Looking Statements
This news release contains forward-looking statements within the meaning of the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. Forward-looking statements include, but are not limited to, statements concerning Zimmer Biomet’s expectations, plans, prospects, and product and service offerings, including new product launches and potential clinical successes. Such statements are based upon the current beliefs and expectations of management and are subject to significant risks, uncertainties and changes in circumstances that could cause actual outcomes and results to differ materially. For a list and description of some of such risks and uncertainties, see Zimmer Biomet’s periodic reports filed with the U.S. Securities and Exchange Commission (SEC). These factors should not be construed as exhaustive and should be read in conjunction with the other cautionary statements that are included in Zimmer Biomet’s filings with the SEC. Forward-looking statements speak only as of the date they are made, and Zimmer Biomet disclaims any intention or obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise. Readers of this news release are cautioned not to rely on these forward-looking statements, since there can be no assurance that these forward-looking statements will prove to be accurate. This cautionary statement is applicable to all forward-looking statements contained in this news release.
References
1 Zhang, Y., et al. Interfacial Frictional Behavior: Cancellous Bone, Cortical Bone, and a Novel Porous Tantalum Biomaterial. Journal of Musculoskeletal Research. 3(4):, 245-251, 1999.
2 Bobyn, J.D., et al. Characteristics of Bone In-growth and Interface Mechanics of a New Porous Tantalum Biomaterial. Journal of Bone and Joint Surgery (British). 81-B(5): 907, 1999.
3 Shirazi-Adl, A., et al. Experimental Determination of Friction Characteristics at the Trabecular Bone / Porous-coated Metal Interface in Cementless Implants. The Journal of Biomedical Research. 27: 167-175, 1993.
4 Internal ZBH estimates
5 Internal Data on File at Zimmer Biomet: ZRR_WA_2409_11. 2011
6 Internal Data on File at Zimmer Biomet ZRR_WA_2403_11. 2011
7 Internal Data on File at Zimmer Biomet ZRR_WA_2551_12. 2012
8 Internal Data on File at Zimmer Biomet ZTR_BI_0133_18. 2018
| Media | Investors | |
| Heather Zoumas-Lubeski +1 445 248 0577 heather.zoumaslubeski@zimmerbiomet.com Melinda de Boer +1 797 7198 melinda.deboer@zimmerbiomet.com | David DeMartino +1 646 531 6115 david.demartino@zimmerbiomet.com Zach Weiner +1 908 591 6955 zach.weiner@zimmerbiomet.com |
SOURCE Zimmer Biomet Holdings, Inc.