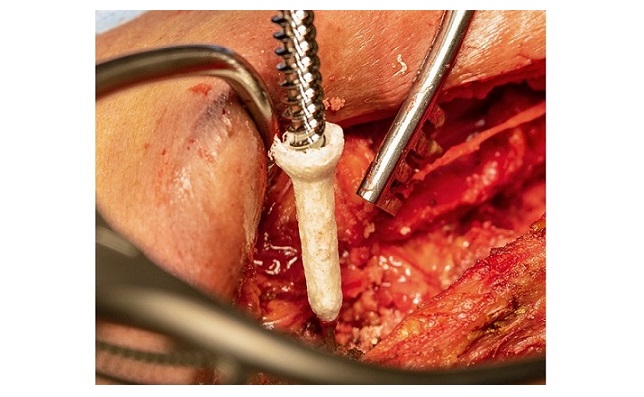
Salt Lake City, UT, June 29th, 2021 – Innovasis, Inc. The Innovasis LxHA® is one of the Industry’s first LLIF devices composed of Hydroxyapatite infused PEEK infused throughout the entire cage. Designed for use in spinal-fusion procedures, the implantable LxHA® lateral interbody contains osteoconductive hydroxyapatite (HA) fully integrated into the matrix of the polymer and exposed on all surfaces of the body of the implant, including within the inner walls of the graft chamber.
This is the fifth PEEK-OPTIMA™© HA Enhanced device marketed by the company. These products, including LxHA®, are available to use in the BioBase™ HA data registry.
The TruView® Lateral Access System was designed to supplement the Innovasis LxHA® PEEK Lateral Intervertebral Body Fusion Device System, which is intended for use in patients with degenerative disc disease at one or two contiguous levels of the lumbar spine (L2-S1).
The TruView® Lateral Access System is a minimally invasive, 2 blade retractor featuring a compact body with radiolucent Carbon Fiber PEEK construction and Aluminum retractor blades for industry leading radiolucency. TruView® has the versatility to add additional A/P blades and can be mounted to the patient or to a table mount.
About Innovasis
Innovasis, Inc. is a rapidly growing company engaged in the research, development, manufacturing, and marketing of spinal implant devices and related products. Innovasis offers a spinal product line with implants and instruments that address the major pathologies and focus areas of traditional spinal surgery. Innovasis is fully committed to providing surgeons and distributors with training, support and excellent customer service, thus ensuring the establishment of a strong and long-term strategic partnership.
Image: Innovasis