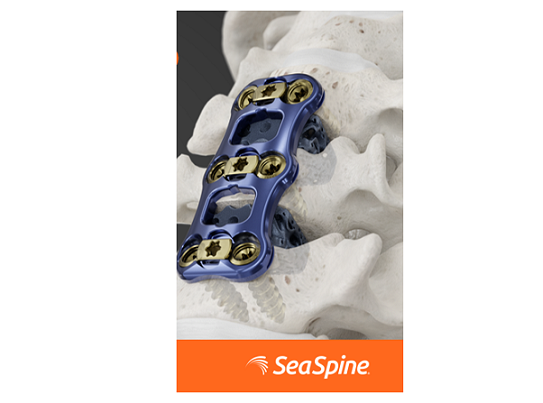
FREDERICK, Md., Nov. 19, 2021 /PRNewswire/ — Theradaptive, a privately held biopharmaceutical company pioneering a new class of regenerative implants announced that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Medical Device designation to its Osteo-Adapt SP Spinal Fusion implant indicated for transforaminal lumbar interbody spinal fusion to treat degenerative disc disease, spondylolisthesis, or retrolisthesis.
The Breakthrough Devices Program is a program for select medical devices that provide for more effective treatment or diagnosis of life-threatening or irreversibly debilitating diseases or conditions. Under the program, the FDA will provide Theradaptive with priority review and interactive communication regarding device development and clinical trial protocols during the premarket review process.
“Theradaptive has developed a technology that may provide several significant advantages over the current state-of-the-art in spinal fusion and musculoskeletal repair, including greater safety and more consistent outcomes,” said Dr. George Muschler of the Cleveland Clinic. “This is an exciting development in the field and having a Breakthrough Designation from the FDA is strong validation of the approach. The potential for patients is enormous.”
Theradaptive’s regenerative implants are best-in-class and are designed to permit precise tissue regeneration for a variety of tissue types and clinical indications. The new technology enables superior outcomes and safety, and greatly improved ease of use.
“This designation is a major milestone for Theradaptive as we aspire to change the practice of medicine by enabling precise tissue regeneration in a way that was not previously possible. This opens the door to entirely new treatments for conditions that currently have very few options,” said John Greenbaum, Senior Vice President of Clinical & Regulatory Operations at Theradaptive.
About Precision Regenerative Implants:
As of 2021 there are no precision regenerative implants available to treat spinal fusion, orthopedic repair, or craniomaxillofacial repair. The current state of the art is based on technology that is over 20 years old and has several serious safety issues. The annual cost of revision procedures in these clinical areas represents a significant burden to the healthcare system.
About Theradaptive:
Theradaptive is a venture-backed biopharmaceutical company developing a therapeutic delivery platform technology for targeted tissue regeneration with a total addressable market of over $10B in several clinical indications. Theradaptive was spun out of the Massachusetts Institute of Technology and has received significant research and development support from the US Department of Defense. Theradaptive has operations in Frederick, MD and Boston, MA.
For more information on Theradaptive, please visit https://www.theradaptive.com
Contact: Serena Lertora; serena.lertora@theradaptive.com
SOURCE Theradaptive