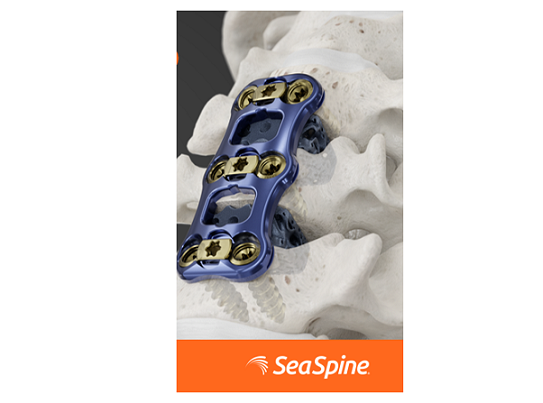
CARLSBAD, Calif., Nov. 23, 2021 (GLOBE NEWSWIRE) — SeaSpine Holdings Corporation (NASDAQ: SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, today announced the full commercial launch of its Admiral ACP System. Admiral ACP represents the next generation of anterior cervical plating and was designed to strike the optimal balance between strength, profile and construct rigidity. Differentiated plate features, combined with step-eliminating innovative instrumentation, work seamlessly in the surgeon’s hands to create a more efficient and consistent anterior cervical discectomy and fusion (ACDF) experience.
Dr. Don Park, Vice Chair of Quality and Safety for the Department of Orthopaedic Surgery at UCLA, said: “Admiral is the most versatile and robust ACDF system I have ever used. The screws, which feature a thread form optimized specifically for high angulation trajectories, and the threaded driver, are game changers. Admiral allows me to tackle my most difficult cases with confidence. I am excited that, with this full commercial launch, the greater market will now have access to such a comprehensive system.”
Admiral ACP is designed to be used as fixation for ACDF procedures in conjunction with the Company’s interbody offerings, including the 3D-printed Waveform™ C and Shoreline® RT, featuring NanoMetalene® surface technology and Reef Topography™. Admiral ACP includes 1-5 level plating options with multiple crossover sizes, which should enable surgeons to intraoperatively address specific surgical needs.
“Our next generation Admiral ACP addresses the largest segment in the estimated $1.3 billion cervical spine surgery market and we believe this system will be a significant growth driver for SeaSpine,” stated Keith Valentine, President and CEO. “This is one more example of SeaSpine’s commitment to launch best-in-class spinal implant systems.”
About SeaSpine
SeaSpine (www.seaspine.com) is a global medical technology company focused on the design, development and commercialization of surgical solutions for the treatment of patients suffering from spinal disorders. SeaSpine has a comprehensive portfolio of orthobiologics and spinal implants solutions to meet the varying combinations of products that neurosurgeons and orthopedic spine surgeons need to perform fusion procedures on the lumbar, thoracic and cervical spine. SeaSpine’s orthobiologics products consist of a broad range of advanced and traditional bone graft substitutes that are designed to improve bone fusion rates following a wide range of orthopedic surgeries, including spine, hip, and extremities procedures. SeaSpine’s spinal implants portfolio consists of an extensive line of products to facilitate spinal fusion in degenerative, minimally invasive surgery (MIS), and complex spinal deformity procedures. Expertise in both orthobiologic sciences and spinal implants product development allows SeaSpine to offer its surgeon customers a differentiated portfolio and a complete solution to meet their fusion requirements. SeaSpine currently markets its products in the United States and in approximately 30 countries worldwide through a committed network of increasingly exclusive distribution partners.
Forward-Looking Statements
SeaSpine cautions you that statements included in this news release that are not a description of historical facts are forward-looking statements that are based on the Company’s current expectations and assumptions. Such forward-looking statements include, but are not limited to, statements relating to: the objectives of product design and the ability of the underlying products to achieve design objectives, including to strike the optimal balance between strength, profile and construct rigidity; whether Admiral ACP addresses the largest segment in the cervical spine surgery market; and whether Admiral ACP will be a significant growth drive for SeaSpine. Among the factors that could cause or contribute to material differences between the Company’s actual results and the expectations indicated by the forward-looking statements are risks and uncertainties that include, but are not limited to: the ability of newly launched products to perform as designed and intended and to meet the needs of surgeons and patients, including as a result of the lack of clinical validation of products in limited commercial (or “alpha”) launch; unexpected delay, including as a result of developing and supporting the launch of new products, including as a result of obtaining regulatory clearances; and other risks and uncertainties more fully described in the Company’s news releases and periodic filings with the Securities and Exchange Commission. The Company’s public filings with the Securities and Exchange Commission are available at www.sec.gov.
You are cautioned not to place undue reliance on forward-looking statements, which speak only as of the date when made. SeaSpine does not intend to revise or update any forward-looking statement set forth in this news release to reflect events or circumstances arising after the date hereof, except as may be required by law.
Investor Relations Contact
Leigh Salvo
(415) 937-5402
ir@seaspine.com