Study shows 57.1% of patients discontinued or reduced oral opioid prescription refills post-procedure and exhibited a decrease in payer costs¹
DUBLIN, Dec. 13, 2021 /PRNewswire/ — Medtronic plc (NYSE: MDT), a global leader in healthcare technology, today announced the results of a study published in Osteoporosis International that demonstrate a decrease, and in some cases elimination of oral opioids, and decrease in payer costs following balloon kyphoplasty (BKP) or vertebroplasty (VP) procedures in patients suffering from spinal fractures, known as vertebral compression fractures (VCF).1 This study, sponsored by Medtronic, is the first published study to evaluate the effect of BKP and VP on opioid reduction as a primary outcome.
An estimated 800,000 VCFs occur in the United States every year.2 They are associated with debilitating pain, spinal deformity, increased mortality, and increased healthcare-resource utilization in elderly patients.3-5 VCFs are often treated with conservative medical management (CMM), including bed rest, back bracing, and prescription oral opioids. These treatments have been shown in clinical studies to have limited effectiveness in reducing pain and improving function long-term compared to interventional treatments like BKP and VP.6-10
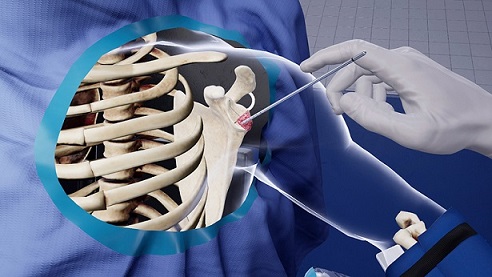
Both BKP and VP treat painful pathological fractures of the vertebral body due to osteoporosis, cancer, or benign lesions. The goal of BKP and VP is to relieve pain and stabilize VCFs. BKP is supported by a robust body of evidence showing important clinical benefits compared to conventional medical management, which include better pain relief and improved quality of life and function.6,7,8,9 These procedures have also been shown to reduce the need for prescription opioids.8
This study, titled, “Trends in opioid use following balloon kyphoplasty or vertebroplasty for the treatment of vertebral compression fractures,” evaluated the effect of BKP or VP on post-procedure opioid prescription fills and payer costs in patients with VCF through a retrospective claims analysis of a large, nationally representative insurance-claims database. Clinical characteristics, opioid-prescription patterns, and payer costs for subjects who underwent either BKP or VP to treat VCF were evaluated beginning six months prior to surgery through seven-months follow-up that included a 30-day post-operative medication washout period to account for the management of post-surgical pain.
The analysis found that 57.1% of patients decreased use of oral opioids, with 48.7% discontinuing use altogether and 8.4% reducing prescription refills post-procedure when compared to the six months prior to the procedure. Additionally, patients who decreased or discontinued opioid prescription fills post-procedure significantly reduced total payer costs over six-month follow-up relative to baseline, with average savings of $6,759.1 Conversely, patients who maintained, increased, or newly started opioids in follow-up slightly increased their payer costs ($160) relative to baseline.
“Our observations are consistent with the existing body of evidence showing a decrease in opioid use following balloon kyphoplasty or vertebroplasty procedures to treat spinal fractures, said Dr. Joshua Hirsch, corresponding author and practicing physician at Massachusetts General Hospital. “Previous studies drew their conclusions from small study populations and clinical trials that were not necessarily designed to measure opioid use as a primary outcome. This study is unique in that it evaluates the impact of these procedures on opioid prescription fills as primary outcome, using a larger, real-world study population.”
“Given the current opioid epidemic and ongoing pain management crisis, we believe there is a need to better explore solutions that effectively address pain and support the elimination of oral opioids,” said Anu Codaty, vice president and general manager, Interventional Pain within the Neuromodulation business, which is part of the Neuroscience Portfolio at Medtronic. “Based on the findings of this study, there may be an added benefit for this patient population in reducing opioid-based harms.”
Medtronic has more than a 40-year history of developing innovative medical devices that have been shown to alleviate pain in different disease states, including BKP, VP, Spinal Cord Stimulation and Targeted Drug Delivery.12 These therapies may help patients manage their pain without the use of oral opioids.
About Medtronic
Bold thinking. Bolder actions. We are Medtronic. Medtronic plc, headquartered in Dublin, Ireland, is the leading global healthcare technology company that boldly attacks the most challenging health problems facing humanity by searching out and finding solutions. Our Mission — to alleviate pain, restore health, and extend life — unites a global team of 90,000+ passionate people across 150 countries. Our technologies and therapies treat 70 health conditions and include cardiac devices, surgical robotics, insulin pumps, surgical tools, patient monitoring systems, and more. Powered by our diverse knowledge, insatiable curiosity, and desire to help all those who need it, we deliver innovative technologies that transform the lives of two people every second, every hour, every day. Expect more from us as we empower insight-driven care, experiences that put people first, and better outcomes for all. In everything we do, we are engineering the extraordinary. For more information on Medtronic (NYSE:MDT), visit www.Medtronic.com and follow @Medtronic on Twitter and LinkedIn.
Any forward-looking statements are subject to risks and uncertainties such as those described in Medtronic’s periodic reports on file with the Securities and Exchange Commission. Actual results may differ materially from anticipated results.
References
- Ni, W., Ricker, C., Quinn, M., Gasquet, N., Janardhanan, D., Gilligan, C. J., & Hirsch, J. A. (2021). Trends in opioid use following balloon kyphoplasty or vertebroplasty for the treatment of vertebral compression fractures. Osteoporosis International.
- Medtronic Data on File. Business Insights & Analytics, September 2019.
- Brunton S, Carmichael B, Gold D, et al. Vertebral compression fractures in primary care: recommendations from a consensus panel. J Fam Pract. 2005;54(9):781-788.
- Ong KL, Beall DP, Frohbergh M, Lau E, Hirsch JA, et al. Were VCF patients at higher risk of mortality following the 2009 publication of the vertebroplasty “sham” trials? Osteoporosis Int. 2018;29(2):375–383. doi:10.1007/s00198-017-4281-z
- Burge R, Dawson-Hughes B, Solomon DH, Wong JB, King A, Tosteson A. Incidence and economic burden of osteoporosis-related fractures in the United States, 2005-2025. J Bone Miner Res. 2007 Mar;22(3):465-75. doi: 10.1359/jbmr.061113. PMID: 17144789.
- Berenson J, Pflugmacher R, Jarzem P, et al. Balloon kyphoplasty versus non-surgical fracture management for treatment of painful vertebral body compression fractures in patients with cancer: a multicentre, randomised controlled trial. Lancet Oncol. 2011;12(3):225-235.
- Wardlaw D, Cummings SR, Van Meirhaeghe J, et al. Efficacy and safety of balloon kyphoplasty compared with non-surgical care for vertebral compression fracture (FREE): a randomised controlled trial. Lancet. 2009;373(9668):1016-1024.
- Boonen S, Van Meirhaeghe J, Bastian L, et al. Balloon kyphoplasty for the treatment of acute vertebral compression fractures: 2-year results from a randomized trial. J Bone Miner Res. 2011;26(7):1627-1637.
- Van Meirhaeghe J, Bastian L, Boonen S, et al. A randomized trial of balloon kyphoplasty and nonsurgical management for treating acute vertebral compression fractures: vertebral body kyphosis correction and surgical parameters. Spine. 2013 38(12),971-983.
- Klazen C, Lohle P, de Vries J, et al. Vertebroplasty versus conservative treatment in acute osteoporotic vertebral compression fractures (Vertos II): an open-label randomised trial. Lancet. 2010;376(9746):1085-1092.
- Data on file: FREE Study, Opioid Use Internal Calculations
- Deer T. Atlas of implantable therapies for pain management. New York, NY: Springer Science and Business Media, LLC; 2011.
UC202210659EN
| Contacts: | |
| Michelle Claypool | Ryan Weispfenning |
| Public Relations | Investor Relations |
| +1-763-526-9452 | +1-763-505-4626 |
SOURCE Medtronic plc