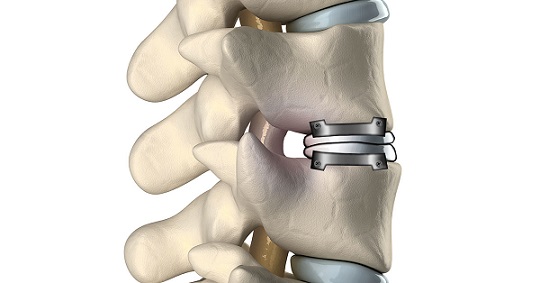
Artificial Cervical Intervertebral Disc Market is projected to reach US$ 4.37529 billion by 2028 from an estimated value of US$ 1.62166 billion in 2022; it is estimated to grow at a CAGR of 18.0% from 2022 to 2028.
New York, July 25, 2022 (GLOBE NEWSWIRE) — The Insight Partners published latest research study on “Artificial Cervical Intervertebral Disc Market Forecast to 2028 – COVID-19 Impact and Global Analysis – by Material, End User, and Geography,” the market Increasing cases of cervical injuries and a rise in the aging population drive the market growth. However, the high cost of cervical intervertebral disc surgeries hampers the artificial cervical intervertebral disc market growth.
Request Sample PDF Copy of Artificial Cervical Intervertebral Disc Market Size – COVID-19 Impact and Global Analysis with Strategic Developments at: https://www.theinsightpartners.com/sample/TIPRE00023194/
Artificial Cervical Intervertebral Disc Market Report Scope & Strategic Insights:
| Report Coverage | Details |
| Market Size Value in | US$ 1.62166 billion in 2022 |
| Market Size Value by | US$ 4.37529 billion by 2028 |
| Growth rate | CAGR of 18.0% from 2022 to 2028. |
| Forecast Period | 2022-2028 |
| Base Year | 2022 |
| No. of Pages | 155 |
| No. Tables | 57 |
| No. of Charts & Figures | 70 |
| Historical data available | Yes |
| Segments covered | Material, End User, and Geography |
| Regional scope | North America; Europe; Asia Pacific; Latin America; MEA |
| Country scope | US, UK, Canada, Germany, France, Italy, Australia, Russia, China, Japan, South Korea, Saudi Arabia, Brazil, Argentina |
| Report coverage | Revenue forecast, company ranking, competitive landscape, growth factors, and trends |
Artificial Cervical Intervertebral Disc Market: Competitive Landscape and Key Developments
Zimmer Biomet Holdings, Inc.; Globus Medical, Inc.; Centinel Spine, LLC; Synergy Spine Solutions, Inc.; B. Braun Melsugen AG; Aditus Medical; AxioMed LLC; NuVasive, Inc.; Medtronic; and Orthofix Medical, Inc. are among the leading companies operating in the artificial cervical intervertebral disc market.
Inquiry Before Purchase: https://www.theinsightpartners.com/inquiry/TIPRE00023194/
Companies operating in the artificial cervical intervertebral disc market implement various organic developments that lead to dynamic improvements in the market. Various organic growth strategies such as product launch and product development propel the market growth. Several companies are implementing numerous inorganic strategies, such as partnerships, and mergers and acquisitions.
In February 2022, AxioMed moved closer toward achieving US FDA approval of its viscoelastic total disc replacement, after submitting PMA module II for the completion of an IDE clinical study. According to AxioMed, the clinical data supporting the device is strong with no reported device-related failures or revisions, and rare cases of heterotopic bone formation.
In September 2021, more than 60,000 M6-C artificial cervical discs of Orthofix Medical were implanted worldwide. The implant is a next-generation artificial disc designed to mimic the natural motion of a native disc. Developed to replace an intervertebral disc damaged by cervical disc degeneration, the M6-C disc is indicated as an alternative to cervical fusion. After the prior approval for distribution under the CE Mark in the European Union and other international geographies, the M6-C disc received an FDA approval in 2019.
In February 2021, NuVasive, Inc., acquired Simplify Medical, the developer of the Simplify Cervical Artificial Disc (Simplify Disc) for cTDR. The acquisition of Simplify Medical added the most clinically effective cTDR technology and distinguished NuVasive’s cervical portfolio in the market. The cervical segment of the global spine market represents an opportunity with an estimated worth of US$ 2.6 billion, and the company has been well-positioned to expand its reach with the addition of the Simplify Disc to its C360 portfolio.
Have a question? Speak to Research Analyst: https://www.theinsightpartners.com/speak-to-analyst/TIPRE00023194
Companies in the artificial cervical intervertebral disc market are undertaking many research and development activities to introduce innovative products. New replacement therapies enable the adoption of advanced technologies, strengthening the procedural outcomes. For instance, in April 2021, US Food and Drug Administration (FDA approved) NuVasive Simplify Cervical Artificial Disc for two-level cervical total disc replacement (cTDR). Additionally, in August 2020, Orthofix Medical Inc., a global medical device company focused on musculoskeletal products and therapies, received the US FDA approval for its M6-C artificial cervical disc offered for treating the patients suffering from cervical disc degeneration. The M6-C artificial cervical disc, a next-generation artificial disc, is developed to replace the damaged intervertebral disc, which also restores physiologic motion to the spine. Moreover, in May 2018, Zimmer Biomet received approval from the Japan Ministry of Health, Labour and Welfare (MHLW) for its Mobi-C Cervical Disc for cervical total disc replacement (cTDR). Mobi-C is the first cervical disc prosthesis approved by the US FDA to reconstruct a cervical disc at one and two levels (C3-C7). Such robust developments are likely to reshape the artificial cervical intervertebral disc market during the forecast period.
In North America, the US holds a significant share of the artificial cervical intervertebral disc market. The market growth in the US is primarily driven by increasing aging population in the US, growing prevalence of cervical degenerative disc disease, and surging number of spine injuries. As per the Fact Sheet: Aging in the United States, the number of individuals in aged 65 and older in the US, is estimated to reach ~95 million by 2060 from 52 million in 2018, and its share in the total population is likely to spike up to 23% from 16%. Further, it is estimated that ~5 million adults in the US are disabled by spine-related disorders; however, candidates for spinal surgery account for a tiny portion of these people. These factors are likely to boost the growth of artificial cervical intervertebral disc market in the US.
Avail Lucrative DISCOUNTS on “Artificial Cervical Intervertebral Disc Market” Research Study: https://www.theinsightpartners.com/discount/TIPRE00023194/
The artificial cervical intervertebral disc market players are adopting various organic and inorganic strategies for market development. For instance, in April 2022, the data of the NuVasive Simplify Cervical Disc (Simplify Disc) two-level US FDA Investigational Device Exemption (IDE) study was published in the Journal of Neurosurgery: Spine. The data will be utilized in the device’s FDA pre-market application for two-level indication approval, which was earlier approved in April 2021
Artificial Cervical Intervertebral Disc Market: Segmental Overview
Based on end user, the artificial cervical intervertebral disc market is segmented into hospitals, ambulatory surgical centers, and others. In 2022, the hospitals segment is estimated to hold the largest share of the market. However, the ambulatory surgical centers segment is expected to experience the highest CAGR during 2022–2028. Ambulatory surgical centers offer cost-effective services in a convenient environment, which is less stressful than many hospitals. They also offer one day procedures, in advanced operating rooms, under the care of highly skilled healthcare professionals. Thus, owing high quality care, cost-effective treatments, and expanding access are likely to favor the market growth of this segment.
Directly Purchase Premium Copy of Artificial Cervical Intervertebral Disc Market Growth Report (2022-2028) at: https://www.theinsightpartners.com/buy/TIPRE00023194/
Browse Adjoining Reports:
Artificial Disc Replacement Systems Market Forecast to 2028 – Covid-19 Impact and Global Analysis – by Disc Type (Composite, Elastic, Mechanical, Others); Location ( Cervical Discs Replacement, Lumbar Disc Replacement ); Material ( Metal, Biopolymer, Mixed, Others ) and Geography
Prestige Cervical Disc Market Forecast to 2028 – COVID-19 Impact and Global Analysis By Product (Metal-on-metal, Metal-on-polymer, Others); End-user (Hospital, Clinic, Others) and Geography
Lumbar Artificial Disc Market Forecast to 2028 – COVID-19 Impact and Global Analysis By Material (Cobalt-chrome Alloys, Titanium Alloys, Stainless Steels, Polyethylene, Polyurethane, Ceramics); End User (Hospitals, Specialty Clinics, Ambulatory Surgical Centers), and Geography
Artificial Disc Market Forecast to 2028 – Covid-19 Impact and Global Analysis – By Type (Cervical, Lumbar); Material (Metal on Biopolymer, Metal on Metal Artificial Disc); End User (Hospitals, Clinics, Homecare Settings, Others)
Disc Prostheses Market Forecast to 2028 – COVID-19 Impact and Global Analysis By Type (Lumbar Disc, Cervical Disc); Application (Metal-on-polymer, Metal-on-metal, Others); and Geography
Motion Preservation Devices Market Forecast to 2028 – Covid-19 Impact and Global Analysis – by Product (Nuclear disc prostheses devices, Annulus repair devices, Artificial discs, Dynamic stabilization devices); Procedure Type (Minimally invasive surgery, Open spine surgery) and Geography
Cervical Vertebra Tractor Market Forecast to 2028 – COVID-19 Impact and Global Analysis by Type (Portable, Mechanical); Application (Household, Hospital, Other)
About Us:
The Insight Partners is a one stop industry research provider of actionable intelligence. We help our clients in getting solutions to their research requirements through our syndicated and consulting research services. We specialize in industries such as Semiconductor and Electronics, Aerospace and Defense, Automotive and Transportation, Biotechnology, Healthcare IT, Manufacturing and Construction, Medical Device, Technology, Media and Telecommunications, Chemicals and Materials.
Contact Us:
If you have any queries about this report or if you would like further information, please contact us:
Contact Person: Sameer Joshi
E-mail: sales@theinsightpartners.com
Phone: +1-646-491-9876
Press Release: https://www.theinsightpartners.com/pr/artificial-cervical-intervertebral-disc-market