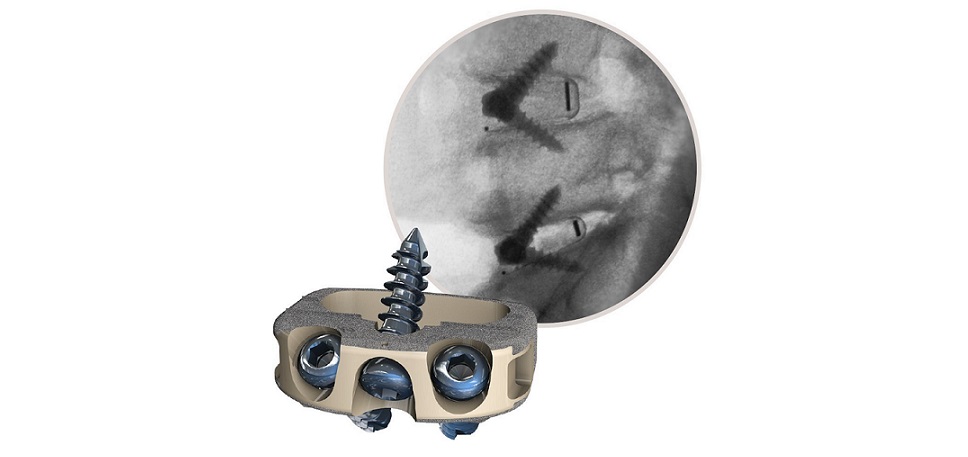
New real-world findings support the effectiveness and use of ReActiv8® Restorative Neurostimulation™ to treat intractable chronic low back pain
November 23, 2022
DUBLIN–( BUSINESS WIRE )– Mainstay Medical Holdings plc today announced the publication of data from a real-world, single-center study with one-year clinical follow-up of patients from the ReActiv8®-C study. Patients who received ReActiv8 implants at Klinikum Itzehoe were sequentially included in this cohort if they had back pain ≥6 and no prior lumbar surgery. The one-year results, published in the journal World Neurosurgery , showed that the majority of the 44 patients who were followed had statistically significant improvements in pain (NRS), disability (ODI), and quality of life (EQ-5D-5L).
This interim analysis suggests that the response to ReActiv8 in these patients is sustained and benefits improve over time, consistent with both a restorative mechanism of action and results from the randomized clinical trial ReActiv8-B.
dr Med. Ardeshir Ardeshiri, Chief Physician, Spine Surgery Section, Trauma Surgery and Orthopedics Clinic, Klinikum Itzehoe, said: “These positive real-world data are consistent with the ReActiv8-B study, which is extremely important to me when introducing new technologies. I look forward to continuing to offer restorative neurostimulation with ReActiv8 to my patients with multifidus dysfunction resulting in chronic axial low back pain.”
Jason Hannon, CEO of Mainstay Medical said , “ These real-world results validate the restorative mechanism of action of ReActiv8, which addresses one of the leading causes of mechanical chronic low back pain, multifidus dysfunction. German physicians are among the early adopters of ReActiv8 and we look forward to making the therapy even more widely available to patients in this region.”
About ReActiv8®
ReActiv8 is an implantable medical device for the treatment of adults with refractory chronic low back pain (CLBP) associated with multifidus muscular dysfunction. Multifidus muscle dysfunction can be detected by imaging or physiological testing in adults who are unresponsive to therapy, including pain relievers and physical therapy, and who are ineligible for spinal surgery. ReActiv8 has received regulatory approval in multiple geographic areas and is commercially available in the European Economic Area, Australia, the UK and the US.
About Mainstay Medical
Mainstay Medical is a medical technology company focused on commercializing its innovative implantable Restorative Neurostimulation™ system, ReActiv8 ® , for patients with disabling mechanical CLBP. Mainstay Medical is headquartered in Dublin, Ireland with subsidiaries in Ireland, USA, Australia, Germany and the Netherlands.
Visit www.mainstaymedical.com for more information.
Forward-Looking Statements
All statements in this release that are not historical facts are, or may be deemed to be, forward-looking statements. These forward-looking statements may include, among other things, statements about the Company’s intentions, beliefs or current expectations regarding the Company’s commercial efforts and performance, financial condition, financing strategies, product design and development, regulatory applications and approvals, and reimbursement agreements.
Forward-looking statements involve risks and uncertainties and are not guarantees of future performance. Actual results could differ materially from those described or implied by the forward-looking statements. A number of factors could cause results and developments to differ materially from those expressed or implied by the forward-looking statements contained herein, including, but not limited to, the risks and uncertainties presented in the Company’s annual report for the year ended December 31, 2021, which is contained in connection with the Company’s public disclosures (available on the Company’s website ( www.mainstaymedical.com) should be read. The forward-looking statements contained herein speak only as of the date of this announcement.
The source language in which the original text is published is the official and authorized version. Translations will be included for a better understanding. Only the language version that was originally published is legally valid. Therefore, compare translations with the original language version of the publication.
Contacts
PR and IR Inquiries:
LifeSci Advisors, LLC
Brian Ritchie
Tel: +1 (212) 915-2578
Email: britchie@lifesciadvisors.com
FTI Consulting (for Ireland)
Jonathan Neilan or Patrick Berkery
Tel : +353 1 765 0886
Email: mainstay@fticonsulting.com
Mainstay Medical
Corporate Communications
Email: Media@mainstaymedical.com