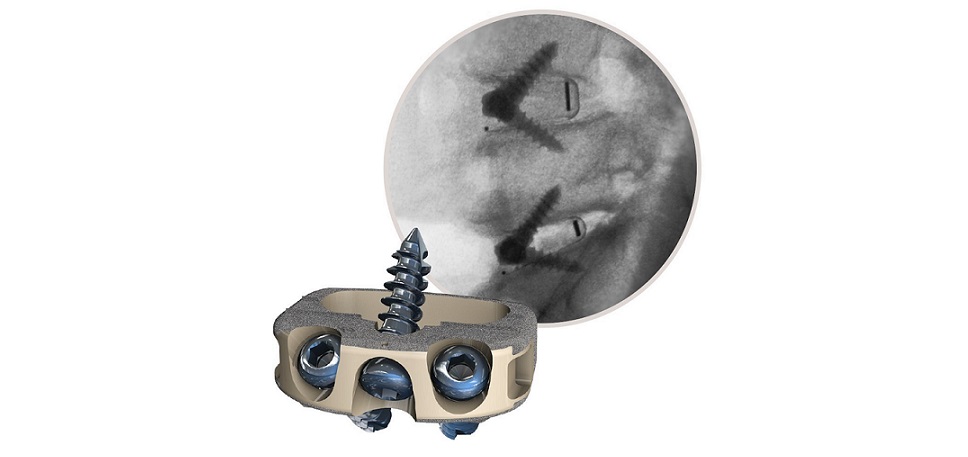
- Study conducted by the Texas Back Institute (TBI), Plano, TX and published in the International Journal of Spine Surgery
- The retrospective study supports utilizing STALIF M portfolio implants as a stand-alone device
- The STALIF technology platform has a proven clinical history of over 30 years and over 75,000 devices implanted
WEST CHESTER, Pa., Nov. 30, 2022 /PRNewswire/ — Centinel Spine®, LLC, (“the Company”) a leading global medical device company addressing cervical and lumbar spinal disease through anterior surgical access, today announced the recent publication of a peer-reviewed study of its STALIF® M portfolio of products in the International Journal of Spine Surgery, titled: “Evaluation of Anterior Lumbar Interbody Fusion Performed Using a Stand-Alone, Integrated Fusion Cage.” 1 The retrospective study, conducted by the Texas Back Institute (TBI), Plano, Texas, supports utilizing STALIF M portfolio implants as a stand-alone device that produces good outcomes in patients treated for symptomatic disc degeneration while avoiding the use of posterior fixation and its complication risk and cost.
“The STALIF M-Ti titanium-coated PEEK cage with integrated screws provides a stable construct, encouraging rapid fusion incorporation. The results of this study further support what I’ve seen clinically for many years, which is good outcomes from stand-alone ALIF for treating painful disc degeneration resistant to non-operative care,” commented Scott L. Blumenthal, MD, TBI.
The STALIF M portfolio of stand-alone integrated cage-screw implants were utilized in a consecutive series of 58 patients with a mean follow-up time of 30.6 months (minimum 24-month follow-up). All available patients’ reported outcome scores demonstrated significant improvement (p<0.01 for all), including Oswestry Disability Index (ODI), visual analogue scale (VAS) back pain, and VAS leg pain. The results of this study corroborate that stand-alone ALIF is a viable procedure for the treatment of symptomatic disc degeneration in patients who have failed nonoperative care and who do not have specific indications for supplemental posterior instrumentation.
“This study is significant because it supports the use of standalone ALIF as a valid treatment option that leads to good clinical outcomes for select patients undergoing spinal fusion,” said Jessica Shellock, MD, TBI. “Standalone ALIF is a very reasonable alternative to consider for patients that don’t require an additional posterior surgery due to instability or severe neural compression and avoids the risks and cost associated with posterior instrumentation.”
The STALIF M implant technology has been engineered based on the STALIF technology platform, which has a proven clinical history of over 30 years and over 75,000 devices implanted2.
“It has a long track record as an anatomic integrated ALIF implant that can be used as a stand-alone device. I like that the STALIF M-Ti has an advanced titanium coating with a modulus of elasticity similar to bone, which minimizes subsidence. I have been extremely happy with its performance,” said Richard D. Guyer, MD, TBI.
“The STALIF M has been my ‘go-to’ standalone ALIF implant-of-choice for many years,” commented Jack Zigler, MD, TBI. “It has a good range of sizes and lordotic angles, and has been consistently hassle-free in using the variable-angle screwdriver and self-tapping screws. After a tough decompression and mobilization, it’s great to know that the implantation itself will be straightforward and will not introduce new problems at the end of the case,” he concluded.
1 Richard D. Guyer, Jack E. Zigler, Scott L. Blumenthal, Jessica L. Shellock and Donna D. Ohnmeiss. Evaluation of Anterior Lumbar Interbody Fusion Performed Using a Stand-Alone, Integrated Fusion Cage. International Journal of Spine Surgery (Aug. 8, 2022). http://ijssurgery.com/content/early/2022/08/07/8354
2 Data on file
About Centinel Spine, LLC
Centinel Spine®, LLC is a leading global medical device company addressing cervical and lumbar spinal disease through anterior surgical access. The company offers a continuum of trusted, brand-name, motion-preserving and fusion solutions backed by over 30 years of clinical success—providing the most robust and clinically-proven technology platforms in the world for total disc replacement (prodisc®) and Integrated Interbody™ fusion (STALIF®).
Centinel Spine continues to advance its pioneering culture and corporate mission to become a catalyst of change in the spine industry and alter the way spine surgery is perceived. Centinel Spine remains the only company with comprehensive motion-preserving and fusion solutions for both cervical and lumbar anterior column reconstruction.
For more information, please visit the company’s website at www.CentinelSpine.com or contact:
Centinel Spine
Varun Gandhi
Chief Financial Officer
900 Airport Road, Suite 3B
West Chester, PA 19380
Phone: 484-887-8871
Email: v.gandhi@centinelspine.com
Media
Kyle Evans
ICR Westwicke
Phone: 646-677-1295
Email: kyle.evans@westwicke.com
SOURCE Centinel Spine, LLC