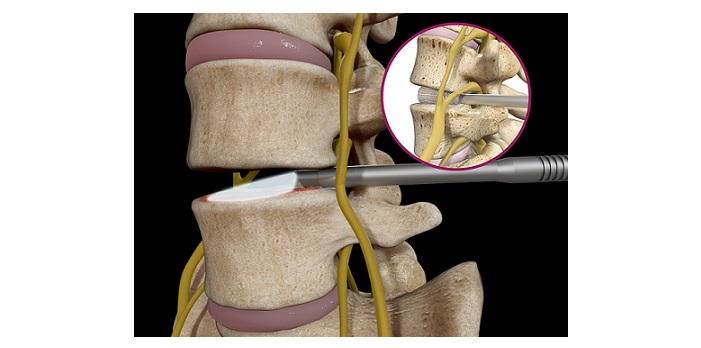
Savannah GA, March 10, 2023 /OrthoSpineNews/ – OC Spine, a division of OC Medical Devices, a privately held medical device company focused on developing, manufacturing, and commercializing spinal implants that address unmet needs, announced the first clinical cases of their recently approved Focus Interbody system featuring the xCELLerate bioactive coating. Cases were performed at The Hospital at Westlake in Austin, Texas by Dr. Daniel Peterson MD, FACS.
“The OC Focus Interbody system using the xCELLerate bioactive coating is a pleasure to use. I am excited to follow the fusion rate in these first clinical cases. The entire OC lumbar spine instrumentation set was easy to use, well-built and gives me great confidence that my patients will have optimal outcomes” stated Dr. Peterson.
Jason Bazemore, Vice President of Sales & Marketing shared “We are thrilled to work with such a skilled clinician as Dr. Peterson and grateful to the staff and administration at The Hospital at Westlake for helping facilitate this launch. As a startup company, feedback from surgeons like Dr. Peterson is how we can react quickly to make changes to our systems before making the substantial investment needed for a full commercial launch. Our xCELLerate bioactive coating is a game changer for us, and we will incorporate it on future product offerings in our development pipeline.”
OC Spine is a division of OC Medical Devices headquartered in Savannah, GA. OC Spine delivers the highest quality spinal implants and instruments that are derived from surgeon input. We develop innovative solutions produced with German manufacturing precision. OC Spine’s goal is to provide sterile implant solutions with the objective of improving patient outcomes.
Media Contact
Jason Bazemore
Vice President of Sales & Marketing
Jason.bazemore@ocmedicaldevices.com
978.489.9020