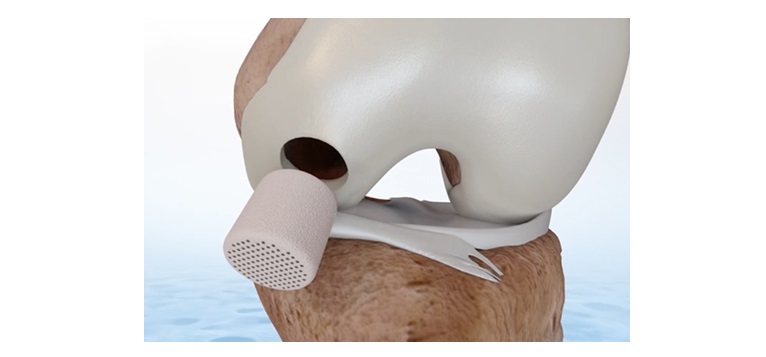
MOVE®-C cervical artificial disc replacement
NONNWEILER, SAARLAND, GERMANY, November 14, 2023 /OrthoSpineNews/ — NGMedical GmbH, a medical device manufacturer exclusively focused on creating innovative technologies for spinal application, announces the first implantation of its MOVE®-C cervical artificial disc replacement in Taiwan.
MOVE®-C combines the features of a second-generation viscoelastic disc prosthesis with the simple implantation technique of a cervical cage. The prosthesis offers physiological motion in all 6 planes including axial damping with progressive resistance to motion. MOVE®-C is the first prosthesis with additively manufactured titanium endplates and avoids the use of PE.
“The first implantation of our MOVE®-C cervical artificial disc replacement in Taiwan marks another important milestone for NGMedical. The market entrance in Taiwan is the next step in the global commercialization of our MOVE®-C artificial disc prosthesis. Taiwan is a very active spine market and we are proud to observe, how MOVE®-C is used and accepted even more by experienced spine surgeons in many countries .” says Peter Weiland, CEO of NGMedical.
About NGMedical
We are an owner-managed company specializing in the development of innovative spinal implants. Many years of experience, combined with a high degree of creativity, give us the impetus to significantly advance the development of spine surgery.
The NGMedical team has been developing innovative product systems since 2001. NGMedical derived out of the company Advanced Medical Technologies AG, which was acquired by Medtronic Inc. in 2012. NGMedical exploits this longtime experience combined with a high degree of creativity and know-how for the development of further innovative product concepts. Both, earlier and today, the cooperation with a team of experienced surgeons and scientists is the basis for the market-driven orientation. Many product solutions, today accepted and widely used as standards, derive from our team.
Peter Weiland
NGMedical GmbH
+49 6873 999970
email us here
Visit us on social media:
Facebook
LinkedIn
Instagram