BOCA RATON, Fla., April 15, 2026 -(BUSINESS WIRE)- SurGenTec, LLC, a leader in innovative spine and orthopedic technologies, today announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for its TiLink™ Navigation Instruments for use with the TiLink™ SI Joint Fusion System in sacroiliac joint procedures. The system is engineered for compatibility with Medtronic’s StealthStation™ navigation platform, enabling enhanced precision during minimally invasive SI joint fusion surgeries.
This milestone expands SurGenTec’s growing portfolio of sacroiliac joint solutions and underscores the company’s commitment to advancing precision-driven technologies that support improved patient outcomes for those suffering from debilitating SI joint pain.
The TiLink™ Navigation instruments were designed to assist surgeons in accurately locating, accessing, and preparing the sacroiliac joint for fusion. When used in conjunction with Medtronic’s StealthStation™, the system enables real-time surgical navigation during implantation of the TiLink™ SI Joint Fusion System. The TiLink implant features proprietary Nanotex® nano-surface technology and a compression-based design intended to support bone integration and joint stabilization. Pre-clinical ovine studies have demonstrated that Nanotex® surface technology, utilizing advanced nanotopography, promotes both bone on-growth and in-growth at the implant interface, supporting enhanced stability and fusion potential.
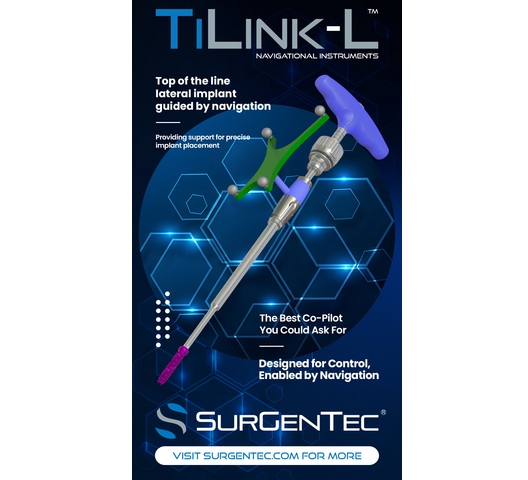
Navigation-assisted techniques are increasingly becoming standard in minimally invasive sacroiliac joint procedures, offering improved accuracy, enhanced visualization, and increased procedural confidence.
Key potential benefits of navigated TiLink™ SI joint fusion include:
- Enhanced precision implant placement and joint compression to support stable fixation
- Improved visualization of complex pelvic anatomy and surrounding critical structures
- Potential reductions in operative time, radiation exposure, and procedural complications
Dr. Joshua Abrams, an orthopedic spine surgeon at the Desert Institute for Spine Care (DISC) in Phoenix, Arizona, stated:
“The TiLink Navigation Instruments introduces a new level of efficiency and control in sacroiliac joint fusion. When used with Navigation, it enables highly accurate, minimally invasive implant placement, which may help reduce operative time while enhancing procedural safety and outcomes for patients with SI joint dysfunction who have not responded to conservative treatment.”
About SurGenTec, LLC
SurGenTec is a privately held medical device company dedicated to advancing spine and orthopedic surgery through innovative, minimally invasive technologies. Focused on improving clinical outcomes and addressing unmet needs in sacroiliac joint treatment, the company offers a comprehensive portfolio that includes the TiLink™ SI Joint Fusion System featuring proprietary Nanotex® nano-surface technology, advanced bone graft solutions, and a range of precision-engineered implants and instruments designed to enhance surgical efficiency and performance. The navigation platform rollout will commence immediately.