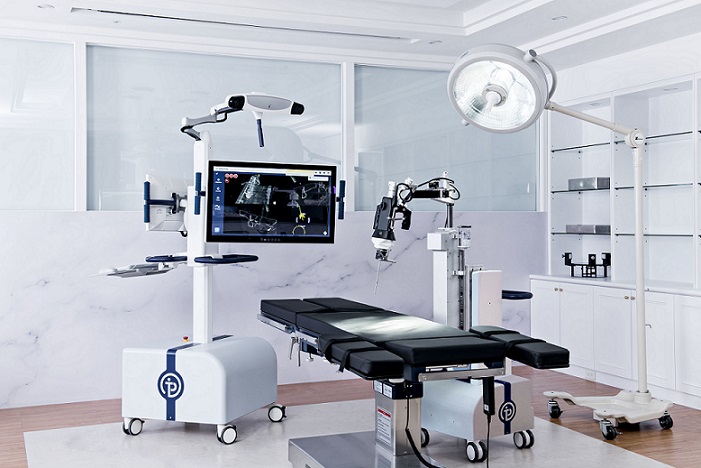
The cervical solution marks the completion of the NuVasive Reline fixation portfolio for complex deformity from occiput to sacrum
SAN DIEGO, Sept. 28, 2022 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally integrated solutions, announced today the commercial launch of Reline® Cervical, a new fixation system for posterior cervical fusion (PCF), in targeted regions.
“Reline Cervical completes the foundational three strategic pillars of C360™—anterior cervical decompression and fusion, cervical total disc replacement, and now PCF. This launch is a significant step forward for our fixation portfolio and furthers our goal of becoming the market leader in complex deformity while extending our position with the industry’s leading cervical portfolio,” said Ryan Donahoe, chief technology officer at NuVasive. “We have leveraged our expertise with the proven Reline system, combined with Pulse®, to deliver our best PCF solution. The key advancements in this procedural technology fuel continued growth and differentiation in the cervical spine segment.”
As cervical spine procedures comprise an approximately $2.6 billion subsegment of the global spine market, the introduction of Reline Cervical will allow the Company’s C360 portfolio to take even more share. Surgeons can address complex PCF surgeries within one comprehensive fixation portfolio, as well as utilize the enabling technologies within Pulse. The compatibility of Reline Cervical with Pulse gives surgeons the benefits of navigation, imaging enhancement, intraoperative neuromonitoring, iGA®, and patient-specific rod bending.
“The Reline Cervical design is best-in-class. I’m impressed by the screw range, great connector options, and the full junctional offering, helping me address the varying complex deformity cases I see in my practice,” said Steven Ludwig, M.D., orthopedic spine surgeon at the University of Maryland. “Having the benefits of the thoracolumbar Reline system now in the cervical spine gives me confidence that I’m giving my patients the best solution.”
The expansion of the C360 portfolio further supports a comprehensive anterior and posterior offering of instrumentation, implants, biologic materials, and enabling technologies for any cervical procedure.
NuVasive will feature its C360 portfolio at these upcoming 2022 events:
- NuVasive Innovation Event: On November 16, join NuVasive virtually to hear Company leadership and surgeon partners discuss all comprehensive procedural portfolios—X360, C360, thoracolumbar posterior, posterior fixation, pediatrics, and the enabling technologies within Pulse. Hear from surgeons on the Reline Cervical system and how it has already made an impact on their practices.
- 50th Cervical Spine Research Society Annual Meeting: From November 16 to 19, visit NuVasive at booth #105/204 to see the full C360 portfolio—Reline Cervical, the Simplify® Cervical Disc, the NuVasive anterior cervical plate system, cervical Advanced Materials Science® interbody implants, and Pulse.
Reline Cervical is available in the U.S. The timeline for availability in other markets cannot be guaranteed.
About NuVasive
NuVasive, Inc. (NASDAQ: NUVA) is the leader in spine technology innovation, with a mission to transform surgery, advance care, and change lives. The Company’s less-invasive, procedurally integrated surgical solutions are designed to deliver reproducible and clinically proven outcomes. The Company’s comprehensive procedural portfolio includes surgical access instruments, spinal implants, fixation systems, biologics, software for surgical planning, navigation and imaging solutions, magnetically adjustable implant systems for spine and orthopedics, and intraoperative neuromonitoring technology and service offerings. With more than $1 billion in net sales, NuVasive operates in more than 50 countries serving surgeons, hospitals, and patients. For more information, please visit www.nuvasive.com.
Forward-Looking Statements
NuVasive cautions you that statements included in this news release that are not a description of historical facts are forward-looking statements that involve risks, uncertainties, assumptions and other factors which, if they do not materialize or prove correct, could cause NuVasive’s results to differ materially from historical results or those expressed or implied by such forward-looking statements. The potential risks and uncertainties which contribute to the uncertain nature of these statements include, among others, risks associated with acceptance of the Company’s surgical products and procedures by spine surgeons and hospitals, development and acceptance of new products or product enhancements, clinical and statistical verification of the benefits achieved via the use of NuVasive’s products, the Company’s ability to adequately manage inventory as it continues to release new products, its ability to recruit and retain management and key personnel, and the other risks and uncertainties described in NuVasive’s news releases and periodic filings with the Securities and Exchange Commission. NuVasive’s public filings with the Securities and Exchange Commission are available at www.sec.gov. NuVasive assumes no obligation to update any forward-looking statement to reflect events or circumstances arising after the date on which it was made.
SOURCE NuVasive, Inc.