April 11, 2023
BORDEAUX, France & BOSTON–(BUSINESS WIRE)–Regulatory News:
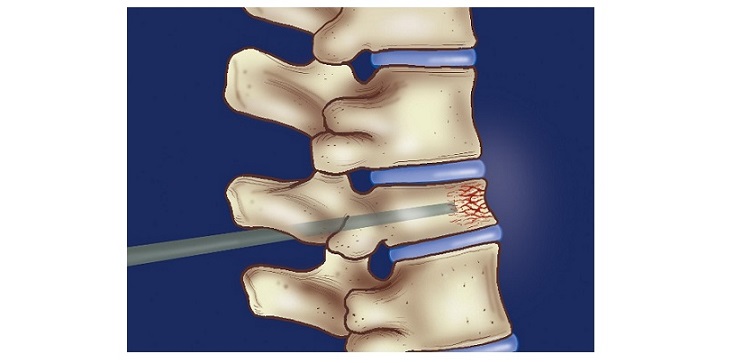
IMPLANET (Euronext Growth: ALIMP, FR0013470168, eligible for PEA-PME equity savings plans) (Paris:ALIMP), a medical technology company specializing in vertebral implants and the distribution of technological medical equipment, today announced the Food and DrugAdministration (FDA) 510(k) clearance of its SqualeTM anterior cervical cage.
The SqualeTM anterior cervical cage, from the OSD product portfolio, features a wide range of implants a design adapted to vertebral anatomy and radiolucent for optimal radiographic confirmation of interbody fusion. To date, more than 15,000 SqualeTM cages have been implanted worldwide, confirming the clinical value of these vertebral fusion devices. The FDA clearance of this product follows the first surgeries in the United States in 2022 with the ORIGINTM cervical plate, which marked the first successful collaboration between Implanet and OSD.
Ludovic Lastennet, IMPLANET’s Chief Executive Officer, stated: “This new clearance demonstrates once again the full potential of the synergies offered by the range of products in the OSD portfolio and is fully in line with our strategy to revitalize our presence in the United States. The Implanet America sales teams should be able to launch the marketing of this anterior cervical cage range before the summer of 2023. This is a new step for our development plan in the United States, aimed in particular at strengthening our offer in the field of spinal surgery in a market estimated at $1.35 billion1.“
Upcoming financial press release
- 2023 First-Quarter Revenue, on April 25, after market close
About IMPLANET
Founded in 2007, IMPLANET is a medical technology company that manufactures high-quality implants for orthopedic surgery and distributing medical technology equipment. Its activity revolves around a comprehensive innovative solution for improving the treatment of spinal pathologies (JAZZ®) complemented by the product range offered by Orthopaedic & Spine Development (OSD), acquired in May 2021 (thoraco-lumbar screws, cages and cervical plates). Implanet’s tried-and-tested orthopedic platform is based on the traceability of its products. Protected by four families of international patents, JAZZ® has obtained 510(k) regulatory clearance from the Food and Drug Administration (FDA) in the United States, the CE mark in Europe and ANVISA approval in Brazil. In 2022, IMPLANET entered into a commercial, technological and financial partnership with SANYOU MEDICAL, China’s second largest medical device manufacturer. IMPLANET employs 43 staff and recorded a consolidated revenue of €8.0 million in 2022. Based near Bordeaux in France, IMPLANET opened a US subsidiary in Boston in 2013. IMPLANET is listed on the Euronext Growth market in Paris. For further information, please visit www.Implanet.com.
————-
1 Source Global Market Model 2018
Contacts
IMPLANET
Ludovic Lastennet, CEO
David Dieumegard, CFO
Tél. : +33 (0)5 57 99 55 55
investors@Implanet.com
NewCap
Investor Relations
Mathilde Bohin
Nicolas Fossiez
Tél.: +33 (0)1 44 71 94 94
Implanet@newcap.eu
NewCap
Media Relations
Arthur Rouillé
Tél.: +33 (0)1 44 71 94 94
Implanet@newcap.eu