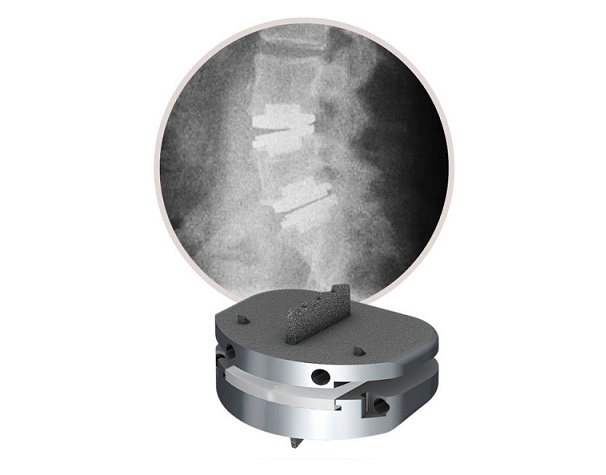
-The American Medical Association announced that it has accepted a new CPT add-on code for the second level of lumbar total disc replacement (TDR) procedures
–Centinel Spine continues to advance lumbar motion preservation, through a two-level FDA approval last year followed by a launch of Angled Endplate implants this year
-Centinel Spine’s prodisc® L is the only total disc replacement system in the U.S. approved for two-level use in the lumbar spine
-Centinel Spine is the only company with FDA-approved cervical and lumbar total disc replacement devices

WEST CHESTER, Pa., Nov. 16, 2021 /PRNewswire/ — Centinel Spine®, LLC, a leading global medical device company addressing cervical and lumbar spinal disease through anterior surgical access, today announced that in an effort to increase the availability of two-level lumbar total disc replacement (TDR) to individuals experiencing degeneration of the intervertebral discs, the American Medical Association (AMA) has accepted the addition of a new add-on Category I Current Procedural Terminology (CPT®) code for a second level of lumbar TDR. Based on published AMA panel action documentation, the effective date for the new CPT code is January 2023, at which time providers can utilize the code when performing two-level lumbar total disc arthroplasty via an anterior approach.
A specific code for a second level lumbar total disc replacement was granted after a thorough review and validation that the technology met the criteria for a Category I CPT code. A critical requirement as part of the review process was high-quality published evidence demonstrating the clinical efficacy of two-level lumbar disc replacement procedures. The CPT codes created and maintained by the AMA CPT Editorial Panel are widely used by third party payors, including Medicare, Medicaid, and commercial health plans.
According to James J. Yue, MD and Morgan Lorio, MD, of the ISASS Coding and Reimbursement Committee, “The International Society for the Advancement of Spine Surgery (ISASS), working in collaboration with regulatory and industry partners, is pleased to announce the successful submission and approval of an AMA Category I CPT code for second level lumbar disc replacement. The approval is a culmination of more than 25 years of motion-sparing scientific, educational, and regulatory activities of ISASS and its physician and industry partners.”
Centinel Spine’s CEO, Steve Murray, stated, “Approval for a second level-specific CPT code is a critical milestone for lumbar total disc arthroplasty and reimbursement plays such a major role in whether patients have access to life-changing technologies. The leadership work of medical societies that support the process for reimbursement code development for patient access and benefit is essential and very much appreciated.”
Jack Zigler, MD from the Texas Health Center for Diagnostics and Surgery, Texas Back Institute, Plano, TX said of the new CPT code for second level TDR, “I am excited for the opportunity to increase patient access to a procedure that has a strong record of success. Patients with two-level lumbar total disc replacements have done remarkably well in long-term follow-up. I have now seen multiple two-level disc replacement patients with over 15-year follow-up who remain positive with their clinical results.”
Centinel Spine continues to innovate in the total disc replacement market, with several recent significant prodisc® L system achievements, including FDA approval of two-level lumbar indications, the launch of new Angled Endplates designed to shift the lordotic angle of the lumbar implant to the inferior endplate, a 2021 Orthopedics This Week Spine Technology Award, and continued advancement of clinical evidence supported through a recent long-term safety and effectiveness study presented during the Best Paper session at the North American Spine Society (NASS) 36th Annual Meeting in September 2021.
About Centinel Spine, LLC
Centinel Spine®, LLC is a leading global medical device company addressing cervical and lumbar spinal disease through anterior surgical access. The company offers a continuum of trusted, brand-name, motion-preserving and fusion solutions backed by over 30 years of clinical success—providing the most robust and clinically-proven technology platforms in the world for total disc replacement (prodisc®) and Integrated Interbody™ fusion (STALIF®).
Centinel Spine continues to advance its pioneering culture and corporate mission to become a catalyst of change in the spine industry and alter the way spine surgery is perceived. Centinel Spine remains the only company with comprehensive motion-preserving and fusion solutions for both cervical and lumbar anterior column reconstruction.
For more information, please visit the company’s website at www.CentinelSpine.com or contact:
Varun Gandhi
Chief Financial Officer
900 Airport Road, Suite 3B
West Chester, PA 19380
Phone: 484-887-8871
Email: v.gandhi@centinelspine.com
SOURCE Centinel Spine, LLC